An adult with an incomplete vaccination history presents with an uncontrollable, rapid and violent cough, fever, and runny nose. Healthcare personnel should suspect
Pertussis.
Rhinovirus.
Bronchitis.
Adenovirus.
The correct answer is A, "Pertussis," as healthcare personnel should suspect this condition based on the presented symptoms and the patient’s incomplete vaccination history. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, pertussis, caused by the bacterium Bordetella pertussis, is characterized by an initial phase of mild respiratory symptoms (e.g., runny nose, low-grade fever) followed by a distinctive uncontrollable, rapid, and violent cough, often described as a "whooping" cough. This presentation is particularly concerning in adults with incomplete vaccination histories, as the pertussis vaccine’s immunity (e.g., DTaP or Tdap) wanes over time, increasing susceptibility (CBIC Practice Analysis, 2022, Domain I: Identification of Infectious Disease Processes, Competency 1.1 - Identify infectious disease processes). Pertussis is highly contagious and poses a significant risk in healthcare settings, necessitating prompt suspicion and isolation to prevent transmission.
Option B (rhinovirus) typically causes the common cold with symptoms like runny nose, sore throat, and mild cough, but it lacks the violent, paroxysmal cough characteristic of pertussis. Option C (bronchitis) may involve cough and fever, often due to viral or bacterial infection, but it is not typically associated with the rapid and violent cough pattern or linked to vaccination status in the same way as pertussis. Option D (adenovirus) can cause respiratory symptoms, including cough and fever, but it is more commonly associated with conjunctivitis or pharyngitis and does not feature the hallmark violent cough of pertussis.
The suspicion of pertussis aligns with CBIC’s emphasis on recognizing infectious disease patterns to initiate timely infection control measures, such as droplet precautions and prophylaxis for exposed individuals (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). Early identification is critical, especially in healthcare settings, to protect vulnerable patients and staff, and the incomplete vaccination history supports this differential diagnosis given pertussis’s vaccine-preventable nature (CDC Pink Book: Pertussis, 2021).
There are four cases of ventilator-associated pneumonia in a surgical intensive care unit with a total of 200 ventilator days and a census of 12 patients. Which of the following BEST expresses how this should be reported?
Ventilator-associated pneumonia rate of 2%
20 ventilator-associated pneumonia cases/1000 ventilator days
Postoperative pneumonia rate of 6% in SICU patients
More information is needed regarding ventilator days per patient
The standard way to report ventilator-associated pneumonia (VAP) rates is:

Why the Other Options Are Incorrect?
A. Ventilator-associated pneumonia rate of 2% – This does not use the correct denominator (ventilator days).
C. Postoperative pneumonia rate of 6% in SICU patients – Not relevant, as the data focuses on VAP, not postoperative pneumonia.
D. More information is needed regarding ventilator days per patient – The total ventilator days are already provided, so no additional data is required.
CBIC Infection Control Reference
APIC and NHSN recommend reporting VAP rates as cases per 1,000 ventilator days.
Which of the following study designs provides the STRONGEST evidence of a causal relationship between a risk factor and the outcome of interest?
Randomized clinical trial
Case-control study
Cohort study
Cross-sectional study
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies the randomized clinical trial (RCT) as the study design that provides the strongest evidence of a causal relationship between a risk factor (or intervention) and an outcome. RCTs are considered the gold standard because they use random assignment to allocate participants to either an intervention group or a control group, which minimizes bias and balances both known and unknown confounding variables between groups.
By controlling exposure and randomly assigning participants, RCTs establish temporality, ensuring that the exposure precedes the outcome—an essential criterion for causality. This design also allows for direct comparison of outcomes under controlled conditions, making it possible to attribute observed differences in outcomes to the intervention or risk factor with a high degree of confidence.
In contrast, cohort studies and case-control studies are observational and can identify associations but are more susceptible to confounding and bias. While cohort studies can demonstrate temporal relationships and estimate risk, they cannot control exposures as precisely as RCTs. Case-control studies are particularly vulnerable to recall and selection bias. Cross-sectional studies assess exposure and outcome simultaneously and cannot establish causation.
For the CIC® exam, it is critical to recognize that randomized clinical trials offer the highest level of evidence for causality, particularly when evaluating interventions, preventive measures, or treatment effectiveness in infection prevention and healthcare epidemiology.
Endemic infection rate refers to
Disease that overwhelms the usual healthcare system.
The usual presence of a disease in a specific population or geographical area.
An outbreak of disease much greater than expected in a specific population or group.
The occurrence of more cases of a disease than expected in a given area or among a specific group.
The CBIC Certified Infection Control Exam Study Guide (6th edition) defines endemic infection rate as the constant or usual presence of a disease within a specific population, geographic area, or healthcare setting. An endemic level represents the baseline or expected frequency of disease occurrence over time, allowing infection preventionists to distinguish normal disease patterns from unusual increases that may signal outbreaks or epidemics.
Option B accurately reflects this definition by describing the expected and stable presence of a disease within a defined population or location. Endemic infections may persist at low or predictable levels and do not necessarily indicate a failure of infection prevention practices. Examples include seasonal influenza in the community or baseline rates of certain healthcare-associated infections within a facility.
Option A refers to a pandemic or healthcare system overload, not endemic disease. Options C and D describe outbreaks or epidemics, which involve a sudden increase in cases above the expected endemic level. These terms imply deviation from baseline and require investigation and intervention.
Understanding endemic rates is critical for infection prevention and surveillance because they provide the comparison point for identifying trends, clusters, and outbreaks. Surveillance data are interpreted against endemic baselines to determine whether changes reflect random variation or meaningful increases requiring action.
For the CIC® exam, recognizing epidemiologic terminology is essential. Endemic infection rate specifically refers to the usual or expected presence of disease, making option B the correct answer.
A nutrition support team wants to determine whether patients who receive total parenteral nutrition (TPN) at home are at increased risk of central line–associated bloodstream infection (CLABSI) compared with patients who receive TPN in the hospital. The BEST way to compare these two groups is to calculate the:
Percentage of patients in each group who became infected.
Infections per 1,000 central line days in each group.
Number of infections in each group this year compared to last year.
Ratio of infected to noninfected central lines in each group.
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that accurate comparison of healthcare-associated infection risk between groups requires use of standardized, exposure-based rates. For central line–associated bloodstream infections (CLABSIs), the recommended metric is infections per 1,000 central line days, which accounts for the amount of time patients are actually exposed to the risk factor—in this case, the presence of a central venous catheter.
Patients receiving TPN at home and those receiving TPN in the hospital may differ substantially in duration of catheter use, care practices, and patient acuity. Simply comparing percentages or raw numbers of infections fails to adjust for differences in central line utilization and can result in misleading conclusions. By using central line days as the denominator, infection rates are normalized and allow for valid comparisons between populations and settings.
Option A does not account for differences in exposure time. Option C compares different time periods rather than comparing risk between groups. Option D provides a ratio but lacks standardization and is not consistent with accepted surveillance methodology.
The Study Guide reinforces that device-associated infection surveillance—such as CLABSI monitoring—must use device days to assess true risk and guide prevention strategies. Understanding and applying correct epidemiologic measures is a core competency for infection preventionists and a frequently tested concept on the CIC® exam.
==========
Infection Prevention and Control identified a cluster of Aspergillus fumigatus infections in the transplant unit. The infection preventionist (IP) meets with the unit director and Environmental Services director to begin investigation. What information does the IP need from the Environmental Services director?
Date of last terminal clean of the infected patient rooms
Hospital grade disinfectant used on the transplant unit
Use of dust mitigating strategies during floor care
Date of the last cleaning of the fish tank in the waiting room
The correct answer is A, "Date of last terminal clean of the infected patient rooms," as this is the most critical information the infection preventionist (IP) needs from the Environmental Services director to begin the investigation of a cluster of Aspergillus fumigatus infections in the transplant unit. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, Aspergillus fumigatus is an environmental fungus that thrives in areas with poor ventilation, construction dust, or inadequate cleaning, posing a significant risk to immunocompromised patients, such as those in transplant units. A terminal clean—thorough disinfection and cleaning of a patient room after discharge or transfer—is a key infection control measure to eliminate fungal spores and other pathogens (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). Determining the date of the last terminal clean helps the IP assess whether lapses in cleaning schedules or procedures could have contributed to the cluster, guiding further environmental sampling or process improvements.
Option B (hospital grade disinfectant used on the transplant unit) is relevant to the investigation but is secondary; the IP would need to know the cleaning schedule first to contextualize the disinfectant’s effectiveness. Option C (use of dust mitigating strategies during floor care) is important, as Aspergillus spores can be aerosolized during floor maintenance, but this is a specific procedural detail that follows the initial focus on cleaning history. Option D (date of the last cleaning of the fish tank in the waiting room) is unlikely to be a priority unless evidence suggests a direct link to the transplant unit, which is not indicated here; Aspergillus is more commonly associated with air quality and room cleaning rather than fish tanks.
The focus on the date of the last terminal clean aligns with CBIC’s emphasis on investigating environmental factors in healthcare-associated infection (HAI) clusters, enabling the IP to collaborate with Environmental Services to pinpoint potential sources and implement corrective actions (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.2 - Analyze surveillance data). This step is foundational to controlling the outbreak and protecting vulnerable patients.
The sensitivity of a laboratory test is the proportion of persons:
With the disease who have a positive test.
With the disease who have a negative test.
Without the disease who have a positive test.
Without the disease who have a negative test.
Sensitivity describes how well a test correctly identifies people who truly have the disease. It is the proportion of true positives among all people with the disease—i.e., the probability that the test will be positive when the disease is present. CDC training materials describe sensitivity as the ability of a test to correctly identify the presence of disease and connect it to true positives and false negatives, with the standard formula: Sensitivity = TP / (TP + FN).
Therefore, the correct definition is “with the disease who have a positive test” (Option A). Option B describes false negatives (people who have disease but test negative). Option C describes false positives (people without disease who test positive). Option D corresponds to specificity, which is the proportion of people without the disease who test negative (TN / [TN + FP]).
In infection prevention and control, understanding sensitivity is essential when selecting and interpreting screening or diagnostic tests (e.g., for outbreak investigations or surveillance), because low sensitivity increases missed cases (false negatives), potentially allowing ongoing transmission if cases are not recognized promptly.
The infection preventionist observed a caregiver entering a room without performing hand hygiene. The BEST response would be to
post additional signage to remind caregivers to wash before entry.
provide immediate feedback and education to the caregiver.
install hand hygiene dispensers in more convenient areas.
design a unit-based education program.
Immediate feedback is a best practice in behavior correction and performance improvement. In hand hygiene non-compliance, real-time intervention allows for immediate correction, education, and reinforcement of infection prevention policies.
The APIC/JCR Workbook recommends:
“Provide simulation training… that provides immediate feedback—for example, how to properly insert a urinary catheter or perform hand hygiene.” This supports behavior change and staff learning.
The APIC Text emphasizes that real-time, direct feedback is more effective than passive measures like signage or delayed education campaigns.
A healthcare worker experiences a percutaneous exposure to a patient with untreated HIV. The next step is to:
Initiate HIV post-exposure prophylaxis (PEP) within 2 hours.
Wait for HIV test results before starting treatment.
Offer post-exposure prophylaxis only if symptoms develop.
Retest for HIV after 6 months before deciding on PEP.
HIV post-exposure prophylaxis (PEP) should be initiated within 2 hours to be most effective.
Waiting for results (B) delays critical treatment.
PEP should always be offered after high-risk exposure, not only if symptoms develop (C).
Retesting after 6 months (D) is recommended but should not delay PEP initiation.
CBIC Infection Control References:
APIC Text, "Bloodborne Pathogens and PEP," Chapter 11.
When evaluating environmental cleaning and disinfectant products as a part of the product evaluation committee, which of the following is responsible for providing information regarding clinical trials?
Infection Preventionist
Clinical representatives
Environmental Services
Manufacturer representatives
The correct answer is D, "Manufacturer representatives," as they are responsible for providing information regarding clinical trials when evaluating environmental cleaning and disinfectant products as part of the product evaluation committee. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, manufacturers are the primary source of data on the efficacy, safety, and performance of their products, including clinical trial results that demonstrate the disinfectant’s ability to reduce microbial load or prevent healthcare-associated infections (HAIs) (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). This information is critical for the committee to assess whether the product meets regulatory standards (e.g., EPA registration) and aligns with infection prevention goals, and it is typically supported by documentation such as peer-reviewed studies or trial data provided by the manufacturer.
Option A (Infection Preventionist) plays a key role in evaluating the product’s fit within infection control practices and may contribute expertise or conduct internal assessments, but they are not responsible for providing clinical trial data, which originates from the manufacturer. Option B (Clinical representatives) can offer insights into clinical usage and outcomes but rely on manufacturer data for trial evidence rather than generating it. Option C (Environmental Services) focuses on the practical application and cleaning processes but lacks the authority or resources to conduct or provide clinical trial information.
The reliance on manufacturer representatives aligns with CBIC’s emphasis on evidence-based decision-making in product selection, ensuring that the product evaluation committee bases its choices on robust, manufacturer-supplied clinical data (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.5 - Use data to guide infection prevention and control strategies). This approach supports the safe and effective implementation of environmental cleaning products in healthcare settings.
A healthy long-term employee with a history of Bacillus Calmette–Guérin (BCG) vaccination has a Tuberculin Skin Test (TST) result of 7 mm induration. The current Centers for Disease Control and Prevention (CDC) recommendations include which of the following?
Send the employee for a chest x-ray
No further action is required
Repeat the test in 1 to 3 weeks
Refer the employee to a physician for treatment
The CBIC Certified Infection Control Exam Study Guide (6th edition) aligns with CDC guidance regarding interpretation of the tuberculin skin test (TST) in healthcare personnel. For a healthy individual with no known risk factors for tuberculosis, a TST is considered positive only when induration is ≥10 mm. In this scenario, the employee’s TST result of 7 mm induration is negative and does not meet the threshold for latent TB infection.
A prior history of BCG vaccination does not change interpretation criteria in adults. The CDC explicitly recommends that TST results be interpreted regardless of BCG history, as vaccine-related reactivity typically wanes over time and induration should not be attributed to BCG alone. Therefore, a 7 mm reaction in a low-risk, asymptomatic healthcare worker does not require further diagnostic evaluation.
Option A (chest x-ray) is reserved for individuals with a positive TB test or symptoms suggestive of active TB. Option C (repeat testing) is not indicated unless this was part of a two-step baseline test and the first result was negative in a newly hired employee, which is not the case here. Option D is inappropriate because treatment is only considered after confirmed latent TB infection.
For the CIC® exam, it is essential to recognize that no further action is required when TST induration is below the positive threshold for the individual’s risk category, even in those with prior BCG vaccination.
An infection preventionist will know a patient may have a pseudo-infection with a positive urine culture because the patient reports:
Flank pain
No symptoms
Urinary frequency
Costovertebral pain
The CBIC Certified Infection Control Exam Study Guide (6th edition) clearly distinguishes true infection from pseudo-infection and colonization, particularly when interpreting urine culture results. A pseudo-infection refers to a positive laboratory result that does not represent a true clinical infection and is often associated with asymptomatic bacteriuria, specimen contamination, or improper collection.
When a patient has a positive urine culture but reports no urinary or systemic symptoms, the most accurate interpretation is that the finding represents colonization or a pseudo-infection rather than a true urinary tract infection (UTI). The Study Guide emphasizes that UTIs must meet both microbiologic and clinical criteria. Symptoms such as dysuria, urinary frequency, urgency, suprapubic tenderness, flank pain, or costovertebral angle tenderness are required to support a diagnosis of infection.
Options A, C, and D all describe symptoms consistent with upper or lower urinary tract infection and therefore suggest true infection rather than pseudo-infection. In contrast, the absence of symptoms strongly supports a diagnosis of asymptomatic bacteriuria or pseudo-infection, for which antimicrobial treatment is generally not indicated, except in specific populations (e.g., pregnant patients or those undergoing urologic procedures).
Recognizing pseudo-infection is critical for antimicrobial stewardship, prevention of unnecessary antibiotic use, and accurate infection surveillance—key competencies tested on the CIC® exam.
An infection preventionist observing the technologist in sterile processing load the washer-disinfector or washer-decontaminator should expect to see them:
Fill the load with mixed items to maximize efficiency.
Put the heaviest items on the top rack of the washer.
Disassemble instruments and open hinged instruments.
Place fresh surgical instruments from the case cart directly onto the rack.
Proper loading of a washer-disinfector is designed to maximize detergent and water contact with all instrument surfaces and internal features. CDC guidance for cleaning and sterilizing practices specifically notes that hinged instruments should be opened fully and items with removable parts should be disassembled (unless the manufacturer provides validated instructions indicating otherwise). This ensures the cleaning solution can reach high-risk areas such as box locks, joints, and crevices, which are common sites for retained soil and bioburden.
The other options describe practices that can compromise cleaning effectiveness and safety. “Filling the load with mixed items to maximize efficiency” (A) risks improper positioning, shadowing, and inadequate exposure of surfaces to spray action and detergent. Placing the heaviest items on the top rack (B) is contrary to common reprocessing guidance, which generally places heavier sets lower to prevent damage and to support effective spray patterns.
Finally, instruments should not be taken “directly from the case cart” onto the rack (D) without appropriate sorting/preparation and following the device manufacturer’s instructions for use (IFU), including opening, disassembly, and correct placement in trays/baskets. The expected best practice during loading is therefore disassemble instruments and open hinged instruments
Bioterrorism agents are classified according to priority. Which organism is listed as a Category A agent?
Q fever
Smallpox
Influenza
Brucellosis
The Certification Study Guide (6th edition) explains that bioterrorism agents are categorized by the Centers for Disease Control and Prevention (CDC) into Categories A, B, and C based on their potential impact on public health. Category A agents represent the highest priority because they pose a severe threat to national security and public health. These agents are characterized by ease of dissemination or transmission, high mortality rates, potential for major public health impact, and the ability to cause public panic and social disruption.
Smallpox (variola virus) is a classic and well-recognized Category A bioterrorism agent. The study guide emphasizes that although naturally occurring smallpox has been eradicated globally, the virus remains a major concern because the general population lacks immunity, person-to-person transmission is efficient, and outbreaks would require extensive public health response. Smallpox also necessitates strict isolation precautions and rapid vaccination strategies during suspected or confirmed cases.
The other options fall into lower categories. Q fever and brucellosis are classified as Category B agents, as they are moderately easy to disseminate but typically cause lower mortality rates. Influenza, while capable of causing pandemics, is not classified as a bioterrorism Category A agent.
Understanding bioterrorism classifications is essential for infection preventionists, particularly in emergency preparedness, surveillance, and response planning—key knowledge areas emphasized on the CIC exam.
In an outbreak of probable foodborne illness, a group of individuals develops watery diarrhea 48 hours after eating imported shellfish. The MOST likely causative organism is:
Vibrio cholerae
Hepatitis A virus
Staphylococcus aureus
Listeria monocytogenes
The CBIC Certified Infection Control Exam Study Guide (6th edition) highlights that consumption of raw or undercooked shellfish, particularly imported shellfish, is a well-recognized risk factor for waterborne and foodborne Vibrio infections, including Vibrio cholerae. The organism thrives in warm coastal waters and can contaminate shellfish harvested from endemic or inadequately regulated regions.
The clinical presentation described—profuse watery diarrhea occurring approximately 48 hours after exposure—is classic for Vibrio cholerae. The organism produces cholera toxin, which causes rapid fluid secretion into the intestinal lumen, resulting in large-volume watery stools. The incubation period typically ranges from a few hours to five days, making a 48-hour onset highly consistent with this pathogen.
The other options are less likely based on incubation period and symptom profile. Hepatitis A virus has an incubation period of weeks and presents with jaundice rather than acute watery diarrhea. Staphylococcus aureus food poisoning causes rapid onset (1–6 hours) due to preformed toxin and is commonly associated with vomiting. Listeria monocytogenes typically causes invasive disease rather than acute watery diarrhea and has a longer incubation period.
For CIC® exam preparation, recognizing shellfish-associated watery diarrhea with short incubation as characteristic of Vibrio cholerae is essential, particularly in outbreak investigations involving imported seafood.
Which of the following stains is used to identify mycobacteria?
Acid-fast
Gram
Methylene blue
India ink
Mycobacteria, including species such as Mycobacterium tuberculosis and Mycobacterium leprae, are a group of bacteria known for their unique cell wall composition, which contains a high amount of lipid-rich mycolic acids. This characteristic makes them resistant to conventional staining methods and necessitates the use of specialized techniques for identification. The acid-fast stain is the standard method for identifying mycobacteria in clinical and laboratory settings. This staining technique, developed by Ziehl-Neelsen, involves the use of carbol fuchsin, which penetrates the lipid-rich cell wall of mycobacteria. After staining, the sample is treated with acid-alcohol, which decolorizes non-acid-fast organisms, while mycobacteria retain the red color due to their resistance to decolorization—hence the term "acid-fast." This property allows infection preventionists and microbiologists to distinguish mycobacteria from other bacteria under a microscope.
Option B, the Gram stain, is a common differential staining technique used to classify most bacteria into Gram-positive or Gram-negative based on the structure of their cell walls. However, mycobacteria do not stain reliably with the Gram method due to their thick, waxy cell walls, rendering it ineffective for their identification. Option C, methylene blue, is a simple stain used to observe bacterial morphology or as a counterstain in other techniques (e.g., Gram staining), but it lacks the specificity to identify mycobacteria. Option D, India ink, is used primarily to detect encapsulated organisms such as Cryptococcus neoformans by creating a negative staining effect around the capsule, and it is not suitable for mycobacteria.
The CBIC’s "Identification of Infectious Disease Processes" domain underscores the importance of accurate diagnostic methods in infection control, including the use of appropriate staining techniques to identify pathogens like mycobacteria. The acid-fast stain is specifically recommended by the CDC and WHO for the initial detection of mycobacterial infections, such as tuberculosis, in clinical specimens (CDC, Laboratory Identification of Mycobacteria, 2008). This aligns with the CBIC Practice Analysis (2022), which emphasizes the role of laboratory diagnostics in supporting infection prevention strategies.
The cleaning and disinfection process that is appropriate for a particular surgical instrument depends on
all surgical instruments are cleaned and sterilized in the same manner.
instruments contaminated with blood must be bleach cleaned first.
the device manufacturer's written instructions for use.
the policies of the sterile processing department.
The correct answer is C, "the device manufacturer's written instructions for use," as this is the factor that determines the appropriate cleaning and disinfection process for a particular surgical instrument. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, the reprocessing of surgical instruments must follow the specific instructions provided by the device manufacturer to ensure safety and efficacy. These instructions account for the instrument’s material, design, and intended use, specifying the appropriate cleaning agents, disinfection methods, sterilization techniques, and contact times to prevent damage and ensure the elimination of pathogens (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). This is also mandated by regulatory standards, such as those from the Food and Drug Administration (FDA) and the Association for the Advancement of Medical Instrumentation (AAMI), which require adherence to manufacturer guidelines to maintain device integrity and patient safety.
Option A (all surgical instruments are cleaned and sterilized in the same manner) is incorrect because different instruments have unique characteristics (e.g., materials like stainless steel vs. delicate optics), necessitating tailored reprocessing methods rather than a one-size-fits-all approach. Option B (instruments contaminated with blood must be bleach cleaned first) is a misconception; while blood contamination requires thorough cleaning, bleach is not universally appropriate and may damage certain instruments unless specified by the manufacturer. Option D (the policies of the sterile processing department) may guide internal procedures but must be based on and subordinate to the manufacturer’s instructions to ensure compliance and effectiveness.
The emphasis on manufacturer instructions aligns with CBIC’s focus on evidence-based reprocessing practices to prevent healthcare-associated infections (HAIs) and protect patients (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). Deviating from these guidelines can lead to inadequate sterilization or instrument damage, increasing infection risks.
An infection preventionist in the role of educator is teaching risk reduction activities to patients and families. For which of the following groups is the pneumococcal vaccine MOST appropriate?
Asplenic patients
International travelers
Immunocompromised newborns
Patients in behavioral health settings
The pneumococcal vaccine is designed to protect against infections caused by Streptococcus pneumoniae, a bacterium responsible for diseases such as pneumonia, meningitis, and bacteremia. The appropriateness of this vaccine depends on the population's risk profile, particularly their susceptibility to invasive pneumococcal disease (IPD). The Certification Board of Infection Control and Epidemiology (CBIC) highlights the role of infection preventionists as educators in promoting vaccination as a key risk reduction strategy, aligning with the "Education and Training" domain (CBIC Practice Analysis, 2022). The Centers for Disease Control and Prevention (CDC) provides specific guidelines on pneumococcal vaccination, recommending it for individuals at higher risk due to underlying medical conditions or immunologic status.
Option A, asplenic patients, refers to individuals who have had their spleen removed (e.g., due to trauma or disease) or have a nonfunctional spleen (e.g., in sickle cell disease). The spleen plays a critical role in clearing encapsulated bacteria like Streptococcus pneumoniae from the bloodstream. Without a functioning spleen, these patients are at significantly increased risk of overwhelming post-splenectomy infection (OPSI), with pneumococcal disease being a leading cause. The CDC and Advisory Committee on Immunization Practices (ACIP) strongly recommend pneumococcal vaccination, including both PCV15/PCV20 and PPSV23, for asplenic patients, making this group the most appropriate for the vaccine in this context. The infection preventionist should prioritize educating these patients and their families about the vaccine's importance and timing.
Option B, international travelers, may benefit from various vaccines depending on their destination (e.g., yellow fever or typhoid), but pneumococcal vaccination is not routinely recommended unless they have specific risk factors (e.g., asplenia or chronic illness) or are traveling to areas with high pneumococcal disease prevalence. This group is not inherently a priority for pneumococcal vaccination. Option C, immunocompromised newborns, includes infants with congenital immunodeficiencies or other conditions, who may indeed require pneumococcal vaccination as part of their routine immunization schedule (e.g., PCV15 or PCV20 starting at 2 months). However, newborns are generally covered under universal childhood vaccination programs, and the question’s focus on "MOST appropriate" suggests a group with a more specific, elevated risk, which asplenic patients fulfill. Option D, patients in behavioral health settings, may have varied health statuses, but this group is not specifically targeted for pneumococcal vaccination unless they have additional risk factors (e.g., chronic diseases), making it less appropriate than asplenic patients.
The CBIC emphasizes tailoring education to high-risk populations, and the CDC’s Adult and Pediatric Immunization Schedules (2023) identify asplenic individuals as a top priority for pneumococcal vaccination due to their extreme vulnerability. Thus, the infection preventionist should focus on asplenic patients as the group for whom the pneumococcal vaccine is most appropriate.
A patient presents with symptoms of meningitis. Two weeks ago, the patient ate from a cheese and meat tray and developed fever, muscle aches, and nausea the next day. Which of the following infectious agents should an infection preventionist consider as a cause of the patient’s symptoms?
Vibrio cholerae
Campylobacter jejuni
Clostridium perfringens
Listeria monocytogenes
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies Listeria monocytogenes as a significant foodborne pathogen capable of causing invasive disease, including meningitis. Listeria is uniquely associated with ready-to-eat foods, particularly soft cheeses, deli meats, pâtés, and meat trays—making the patient’s dietary history highly suggestive. Unlike many other foodborne organisms, Listeria can grow at refrigeration temperatures, increasing the risk of contamination in processed and stored foods.
The incubation period for invasive listeriosis can range from several days to weeks, which aligns with the timeline described. Early symptoms often include fever, myalgias, nausea, and gastrointestinal upset, followed by progression to meningitis or bloodstream infection, especially in high-risk populations such as older adults, pregnant individuals, neonates, and immunocompromised patients. The study guide emphasizes that Listeria is an important consideration when meningitis follows a compatible food exposure history.
The other organisms listed are primarily associated with self-limited gastrointestinal illness, not meningitis. Vibrio cholerae causes severe watery diarrhea; Campylobacter jejuni causes enteritis; and Clostridium perfringens causes toxin-mediated food poisoning with rapid onset diarrhea and abdominal cramping. None are typical causes of meningitis.
This question highlights a high-yield CIC exam concept: linking food exposure history to invasive pathogens, particularly Listeria monocytogenes, which requires prompt recognition and intervention.
==========
Surgical site infection (SSI) data for the previous quarter reveal the following numbers. The surgeon with the highest infection rate is Doctor
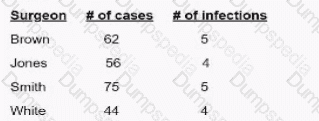
Brown
Jones.
Smith
White
To determine which surgeon has the highest surgical site infection (SSI) rate, use the following formula:
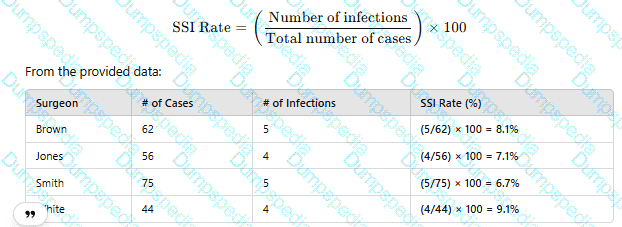
Since Dr. White has the highest SSI rate at 9.1%, the correct answer is D. White.
CBIC Infection Control Reference
SSI rates are calculated using infection count per total procedures and reported as percentage values.
When preparing an educational session, objectives should be written using clear, precise language to describe what the learner is expected to learn. All of the following are examples of appropriate objective verbs EXCEPT:
know
define
compare
explain
The Certification Study Guide (6th edition) emphasizes that educational objectives must be written using measurable, observable action verbs that clearly define learner outcomes. This principle is grounded in adult learning theory and Bloom’s taxonomy, both of which are highlighted in the education and communication sections of the guide. Effective objectives allow educators to evaluate whether learning has occurred by observing or assessing learner performance.
The verb “know” is considered inappropriate because it is vague and not measurable. It does not specify how the learner will demonstrate knowledge, making it impossible to objectively assess whether the objective has been achieved. For this reason, “know” is frequently cited in the study guide as an example of a poorly written objective verb.
In contrast, verbs such as define, compare, and explain are acceptable because they describe specific actions the learner can perform. These verbs allow the instructor to evaluate learning through verbal responses, written assessments, or demonstrations. The study guide stresses that well-written objectives should answer the question: What will the learner be able to do at the end of the session?
This concept is commonly tested on the CIC exam because infection preventionists are expected to design and deliver education effectively. Clear, measurable objectives support competency-based education, documentation of learning outcomes, and program evaluation—all essential components of a successful infection prevention program.
An infection preventionist is educating healthcare personnel on standard precautions and isolation requirements. Sessions consist of large group lectures and demonstrations. Three months later, a carbapenem-resistant Enterobacterales (CRE) outbreak occurs. Which of the following strategies will minimize the risk of a subsequent outbreak?
Reviewing the educational content to ensure that it includes the most recent recommendations
Creating a test to be used at the end of the education sessions to evaluate learning
Recording the lecture to allow staff more time to review the recording when convenient
Meeting with the managers to assess staff compliance with isolation precautions
The Certification Study Guide (6th edition) stresses that education alone is insufficient to ensure sustained adherence to infection prevention practices. While lectures and demonstrations are valuable for knowledge dissemination, they do not guarantee consistent behavioral compliance over time. In this scenario, the occurrence of a CRE outbreak three months after education indicates a gap between knowledge and practice.
To minimize the risk of a subsequent outbreak, the most effective strategy is directly assessing staff compliance with isolation precautions, which is best accomplished by engaging managers and leadership. The study guide emphasizes the importance of monitoring, auditing, and feedback as core components of an effective infection prevention program. Managers are uniquely positioned to observe daily practice, reinforce expectations, and hold staff accountable for adherence to standard and transmission-based precautions.
The other options focus primarily on educational reinforcement rather than practice validation. Updating content, testing knowledge, or offering recorded lectures may improve awareness but do not address whether staff are actually implementing precautions correctly at the point of care. CRE transmission is most often linked to failures in hand hygiene, contact precautions, and environmental cleaning—issues that require ongoing observation and performance management, not passive education.
This question reflects a common CIC exam theme: preventing outbreaks requires behavioral verification and leadership engagement, not education alone. By assessing and reinforcing compliance through managers, the infection preventionist addresses the root cause of transmission risk and supports sustainable prevention.
Which water type is suitable for drinking yet may still be a risk for disease transmission?
Purified water
Grey water
Potable water
Distilled water
To determine which water type is suitable for drinking yet may still pose a risk for disease transmission, we need to evaluate each option based on its definition, treatment process, and potential for contamination, aligning with infection control principles as outlined by the Certification Board of Infection Control and Epidemiology (CBIC).
A. Purified water: Purified water undergoes a rigorous treatment process (e.g., reverse osmosis, distillation, or deionization) to remove impurities, contaminants, and microorganisms. This results in water that is generally safe for drinking and has a very low risk of disease transmission when properly handled and stored. However, if the purification process is compromised or if contamination occurs post-purification (e.g., due to improper storage or distribution), there could be a theoretical risk. Nonetheless, purified water is not typically considered a primary source of disease transmission under standard conditions.
B. Grey water: Grey water refers to wastewater generated from domestic activities such as washing dishes, laundry, or bathing, which may contain soap, food particles, and small amounts of organic matter. It is not suitable for drinking due to its potential contamination with pathogens (e.g., bacteria, viruses) and chemicals. Grey water is explicitly excluded from potable water standards and poses a significant risk for disease transmission, making it an unsuitable choice for this question.
C. Potable water: Potable water is water that meets regulatory standards for human consumption, as defined by organizations like the World Health Organization (WHO) or the U.S. Environmental Protection Agency (EPA). It is treated to remove harmful pathogens and contaminants, making it safe for drinking under normal circumstances. However, despite treatment, potable water can still pose a risk for disease transmission if the distribution system is contaminated (e.g., through biofilms, cross-connections, or inadequate maintenance of pipes). Outbreaks of waterborne diseases like Legionnaires' disease or gastrointestinal infections have been linked to potable water systems, especially in healthcare settings. This makes potable water the best answer, as it is suitable for drinking yet can still carry a risk under certain conditions.
D. Distilled water: Distilled water is produced by boiling water and condensing the steam, which removes most impurities, minerals, and microorganisms. It is highly pure and safe for drinking, often used in medical and laboratory settings. Similar to purified water, the risk of disease transmission is extremely low unless contamination occurs after distillation due to improper handling or storage. Like purified water, it is not typically associated with disease transmission risks in standard use.
The key to this question lies in identifying a water type that is both suitable for drinking and has a documented potential for disease transmission. Potable water fits this criterion because, while it is intended for consumption and meets safety standards, it can still be a vector for disease if the water supply or distribution system is compromised. This is particularly relevant in infection control, where maintaining water safety in healthcare facilities is a critical concern addressed by CBIC guidelines.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain III: Prevention and Control of Infectious Diseases, which highlights the importance of water safety and the risks of contamination in potable water systems.
CBIC Examination Content Outline, Domain IV: Environment of Care, which includes managing waterborne pathogens (e.g., Legionella) in potable water supplies.
Each item or package that is prepared for sterilization should be labeled with the
storage location.
type of sterilization process.
sterilizer identification number or code.
cleaning method (e.g., mechanical or manual).
The correct answer is C, "sterilizer identification number or code," as this is the essential information that each item or package prepared for sterilization should be labeled with. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, proper labeling of sterilized items is a critical component of infection prevention and control to ensure traceability and verify the sterilization process. The sterilizer identification number or code links the item to a specific sterilization cycle, allowing the infection preventionist (IP) and sterile processing staff to track the equipment used, confirm compliance with standards (e.g., AAMI ST79), and facilitate recall or investigation if issues arise (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). This labeling ensures that the sterility of the item can be assured and documented, protecting patient safety by preventing the use of inadequately processed items.
Option A (storage location) is important for inventory management but is not directly related to the sterilization process itself and does not provide evidence of the sterilization event. Option B (type of sterilization process) indicates the method (e.g., steam, ethylene oxide), which is useful but less critical than the sterilizer identification, as the process type alone does not confirm the specific cycle or equipment used. Option D (cleaning method, e.g., mechanical or manual) is a preliminary step in reprocessing, but it is not required on the sterilization label, as the focus shifts to sterilization verification once the item is prepared.
The requirement for a sterilizer identification number or code aligns with CBIC’s emphasis on maintaining rigorous tracking and quality assurance in the reprocessing of medical devices, ensuring accountability and adherence to best practices (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). This practice is mandated by standards such as AAMI ST79 to support effective infection control in healthcare settings.
A positive biological indicator is reported to the Infection Preventionist (IP) after a sterilizer was used. Which of the following should be done FIRST?
Check the Central Services employees' technique
Inform the risk manager of the positive indicator
Notify potentially affected patients of exposure to nonsterile equipment
Re-challenge the sterilizer with a second indicator
When a positive biological indicator (BI) is detected, the immediate response is to retest the sterilizer using another BI to confirm results. This helps distinguish between a true sterilization failure and a defective BI.
The CBIC Study Guide advises:
“If there is no indication of abnormalities, then the sterilizer should be tested again in three consecutive cycles using paired biological indicators from different manufacturers.”
Immediate recall is reserved for implant loads or confirmed sterilization failure.
Incorrect responses:
A. Check employee technique may be appropriate later but not as a first step.
B. Informing risk manager or C. Notifying patients occurs only after confirmation of failure.
The BEST choice for surgical instrument cleaning and material compatibility is a detergent solution with:
An acidic pH
A neutral pH
Sodium hypochlorite
Quaternary ammonium compounds
The Certification Study Guide (6th edition) emphasizes that the primary goal of surgical instrument cleaning is to remove organic and inorganic soil while preserving the integrity and functionality of the instrument. For this reason, detergents with a neutral pH are considered the best choice for routine surgical instrument cleaning and material compatibility.
Neutral pH detergents are effective at removing blood, tissue, and other organic matter without causing corrosion, pitting, or degradation of metals, plastics, seals, and coatings commonly used in surgical instruments. The study guide notes that repeated exposure to harsh chemical environments can damage instruments, compromise device performance, and shorten instrument lifespan—ultimately affecting patient safety and increasing replacement costs.
Acidic detergents may be used selectively for removal of mineral deposits or water scale but are not appropriate for routine cleaning due to their corrosive potential. Sodium hypochlorite (bleach) is strongly discouraged for surgical instruments because it is highly corrosive and can rapidly damage stainless steel. Quaternary ammonium compounds are low-level disinfectants and are not suitable for cleaning critical or semi-critical medical devices prior to disinfection or sterilization.
This question reflects a high-yield CIC exam principle: effective cleaning must balance soil removal with material compatibility. Neutral pH detergents best meet both requirements and are widely recommended by manufacturers and reprocessing standards for surgical instrumentation.
Which of the following process performance indicators should result in improvement in central line–associated bloodstream infections (CLABSI)?
All patients with a central line are on total parenteral nutrition (TPN).
100% compliance with the insertion bundle.
Povidone-iodine antiseptic ointment placed at the insertion site of a peripherally inserted central catheter line.
Routinely changing the central line over a guidewire every seven days.
The Certification Study Guide (6th edition) emphasizes that process performance indicators directly linked to evidence-based practices are the most effective measures for reducing healthcare-associated infections such as CLABSI. Among the options listed, 100% compliance with the central line insertion bundle is the only indicator consistently demonstrated to reduce CLABSI rates.
Insertion bundles are standardized sets of practices that include proper hand hygiene, maximal sterile barrier precautions, use of appropriate skin antisepsis (preferably chlorhexidine), optimal catheter site selection, and daily review of line necessity. The study guide explains that reliable execution of these bundled practices addresses the most common routes of microbial entry at the time of line placement, which is a critical risk period for bloodstream infection.
The other options do not represent valid improvement indicators. Total parenteral nutrition is a known risk factor for CLABSI, not a prevention strategy. Use of povidone-iodine ointment at insertion sites is not recommended and may increase infection risk. Routine guidewire exchanges are discouraged because they do not reduce infection risk and may increase contamination.
Therefore, measuring and achieving full compliance with the insertion bundle is a meaningful, actionable performance indicator that aligns with CBIC exam expectations and infection prevention best practices.
How can infection preventionists BEST educate on a new infection prevention protocol for adoption across different departments within an organization?
Relate relevance of each department’s role using scenarios.
Utilize a passive learning method to accommodate for diversity.
Assign reading and review of protocol with manager sign-off.
Tie protocol compliance metric to annual performance review.
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that effective education for adult learners is most successful when it is relevant, interactive, and role-specific. Relating the new infection prevention protocol to each department’s responsibilities using realistic scenarios is the most effective educational strategy for organization-wide adoption.
Scenario-based education is an active learning method, which engages participants in problem-solving and application of knowledge rather than passive receipt of information. By tailoring scenarios to departmental workflows—such as nursing, environmental services, laboratory, or ancillary departments—staff can clearly understand how the protocol affects their daily practice and how their actions contribute to infection prevention outcomes. This approach improves comprehension, retention, and compliance.
Option B is incorrect because passive learning methods (e.g., lectures or handouts alone) are less effective for behavior change and adult learning. Option C relies on administrative acknowledgment rather than understanding and does not ensure competency or consistent application. Option D may support accountability but does not educate staff or build understanding during initial implementation.
The Study Guide stresses that infection preventionists must act as educators and change agents, adapting teaching strategies to diverse audiences. Using scenario-based, department-specific education aligns with adult learning principles, promotes engagement, and facilitates sustainable practice change—making it the best approach and a key concept for the CIC® exam.
==========
In evaluating the infection control and ventilation measures for operating rooms the Infection Preventionist should know that the air changes per hour (ACH) should be maintained at greater than or equal to 15 ACH. How many of these changes should be fresh air?
Greater than or equal to 3
Greater than or equal to 5
Greater than or equal to 6
Greater than or equal to 7
In operating rooms, a minimum of 15 air changes per hour (ACH) is required, with at least 3 of those ACH being from fresh or outdoor air. This requirement helps reduce microbial contamination and provides a clean surgical environment.
According to the APIC Text:
"In each, air should flow out of the room and the minimum ACH should be 15, with three of these ACH being fresh or outdoor air."
This aligns with design specifications outlined in the 2006 Guidelines for design and construction of health care facilities.
Which statistical test is MOST appropriate for comparing infection rates before and after an intervention?
Student’s t-test
Chi-square test for proportions
Linear regression analysis
Wilcoxon rank-sum test
The Chi-square test is the most appropriate test for comparing infection rates (categorical data) before and after an intervention.
CBIC Infection Control References:
CIC Study Guide, "Statistical Analysis in Infection Control," Chapter 5.
Assume the mean age of onset for patients with tuberculosis (TB) is 62 years, with one standard deviation of 5 years, and the age of onset follows a normal distribution. What is the percentage of patients expected to have the age of onset ranging from 57 to 67 years?
34%
68%
95%
99%
To determine the percentage of patients with an age of onset ranging from 57 to 67 years, we need to apply the properties of a normal distribution. In a normal distribution, the mean represents the central point, and the standard deviation defines the spread of the data. Here, the mean age of onset is 62 years, and the standard deviation is 5 years. The range of 57 to 67 years corresponds to one standard deviation below the mean (62 - 5 = 57) to one standard deviation above the mean (62 + 5 = 67).
In a normal distribution, approximately 68% of the data falls within one standard deviation of the mean (i.e., between μ - σ and μ + σ, where μ is the mean and σ is the standard deviation). This is a well-established statistical principle, often referred to as the 68-95-99.7 rule (or empirical rule) in statistics. Specifically, 34% of the data lies between the mean and one standard deviation above the mean, and another 34% lies between the mean and one standard deviation below the mean, totaling 68% for the range spanning one standard deviation on both sides of the mean.
Let’s verify this:
The lower bound (57 years) is exactly one standard deviation below the mean (62 - 5 = 57).
The upper bound (67 years) is exactly one standard deviation above the mean (62 + 5 = 67).
Thus, the range from 57 to 67 years encompasses the middle 68% of the distribution.
Option A (34%) represents the percentage of patients within one standard deviation on only one side of the mean (e.g., 62 to 67 or 57 to 62), not the full range. Option C (95%) corresponds to approximately two standard deviations from the mean (62 ± 10 years, or 52 to 72 years), which is wider than the given range. Option D (99%) aligns with approximately three standard deviations (62 ± 15 years, or 47 to 77 years), which is even broader. Since the question specifies a range of one standard deviation on either side of the mean, the correct answer is 68%, corresponding to Option B.
In infection control, understanding the distribution of disease onset ages can help infection preventionists identify at-risk populations and allocate resources effectively, aligning with the CBIC’s focus on surveillance and data analysis (CBIC Practice Analysis, 2022). While the CBIC does not directly address statistical calculations in its core documents, the application of normal distribution principles is a standard epidemiological tool endorsed in public health guidelines, which inform CBIC practices.
An infection preventionist is calculating measures of central tendency regarding duration of a surgical procedure using this data set: 2, 2, 3, 4, and 9. Which of the following statements is correct?
The median is 2.
The mode is 3.
The mean is 4.
The standard deviation is 7.
Measures of central tendency (mean, median, mode) and dispersion (standard deviation) are statistical tools used to summarize data, such as the duration of surgical procedures, which can help infection preventionists identify trends or risks for surgical site infections. The Certification Board of Infection Control and Epidemiology (CBIC) supports the use of data analysis in the "Surveillance and Epidemiologic Investigation" domain, aligning with epidemiological principles outlined by the Centers for Disease Control and Prevention (CDC). The question provides a data set of 2, 2, 3, 4, and 9, and requires determining the correct statement by calculating these measures.
Mean: The mean is the average of the data set, calculated by summing all values and dividing by the number of observations. For the data set 2, 2, 3, 4, and 9:(2 + 2 + 3 + 4 + 9) ÷ 5 = 20 ÷ 5 = 4. Thus, the mean is 4, making Option C correct.
Median: The median is the middle value when the data set is ordered. With five values (2, 2, 3, 4, 9), the middle value is the third number, which is 3. Option A states the median is 2, which is incorrect.
Mode: The mode is the most frequently occurring value. In this data set, 2 appears twice, while 3, 4, and 9 appear once each, making 2 the mode. Option B states the mode is 3, which is incorrect.
Standard Deviation: The standard deviation measures the spread of data around the mean. For a small data set like this, the calculation involves finding the variance (average of squared differences from the mean) and taking the square root. The mean is 4, so the deviations are: (2-4)² = 4, (2-4)² = 4, (3-4)² = 1, (4-4)² = 0, (9-4)² = 25. The sum of squared deviations is 4 + 4 + 1 + 0 + 25 = 34. The variance is 34 ÷ 5 = 6.8, and the standard deviation is √6.8 ≈ 2.61 (not 7). Option D states the standard deviation is 7, which is incorrect without further context (e.g., a population standard deviation with n-1 denominator would be √34 ≈ 5.83, still not 7).
The CBIC Practice Analysis (2022) and CDC guidelines encourage accurate statistical analysis to inform infection control decisions, such as assessing surgical duration as a risk factor for infections. Based on the calculations, the mean of 4 is the only correct statement among the options, confirming Option C as the answer. Note that the standard deviation of 7 might reflect a miscalculation or misinterpretation (e.g., using a different formula or data set), but with the given data, it does not hold.
Given the formula for calculating incidence rates, the Y represents which of the following?

Population served
Number of infected patients
Population at risk
Number of events
Incidence rate is a fundamental epidemiological measure used to quantify the frequency of new cases of a disease within a specified population over a defined time period. The Certification Board of Infection Control and Epidemiology (CBIC) supports the use of such metrics in the "Surveillance and Epidemiologic Investigation" domain, aligning with the Centers for Disease Control and Prevention (CDC) "Principles of Epidemiology in Public Health Practice" (3rd Edition, 2012). The formula provided, XY×K=Rate\frac{X}{Y} \times K = RateYX×K=Rate, represents the standard incidence rate calculation, where KKK is a constant (e.g., 1,000 or 100,000) to express the rate per unit population, and the question asks what YYY represents among the given options.
In the incidence rate formula, XXX typically represents the number of new cases (or events) of the disease occurring during a specific period, and YYY represents the population at risk during that same period. The ratio XY\frac{X}{Y}YX yields the rate per unit of population, which is then multiplied by KKK to standardize the rate (e.g., cases per 1,000 persons). The CDC defines the denominator (YYY) as the population at risk, which includes individuals susceptible to the disease over the observation period. Option B ("Number of infected patients") might suggest XXX if it specified new cases, but as the denominator YYY, it is incorrect because incidence focuses on new cases relative to the at-risk population, not the total number of infected individuals (which could include prevalent cases). Option C ("Population at risk") correctly aligns with YYY, representing the base population over which the rate is calculated.
Option A, "Population served," is a broader term that might include the total population under care (e.g., in a healthcare facility), but it is not specific to those at risk for new infections, making it less precise. Option D, "Number of events," could align with XXX (new cases or events), but as the denominator YYY, it does not fit the formula’s structure. The CBIC Practice Analysis (2022) and CDC guidelines reinforce that the denominator in incidence rates is the population at risk, ensuring accurate measurement of new disease occurrence.
An infection preventionist (IP) is asked to evaluate a series of published sources about CAUTI prevention strategies. Which source shows the strongest evidence for the IP to implement change?
A randomized controlled trial from another hospital
Evidence from reports written by authorities who are on expert committees
Data from a case study from a well-designed case-control or cohort study
A systematic review of relevant controlled studies and evidence-based practices
When an IP is selecting evidence to support practice change, the “strength” of evidence is typically judged using an evidence hierarchy. In most evidence pyramids, systematic reviews (often with meta-analysis) of well-designed studies sit at or near the top because they use explicit methods to search for, appraise, and synthesize findings across multiple studies—reducing the influence of chance results and individual-study bias.
Option D is therefore strongest: a systematic review of relevant controlled studies and evidence-based practices provides the most robust overall summary for decision-making compared with any single study. Randomized controlled trials (option A) are strong primary studies, but they represent one setting/population and can be affected by local factors; a high-quality systematic review places RCTs in context and evaluates consistency across multiple trials.
Observational designs (option C, cohort/case-control) are generally lower in the hierarchy for intervention effectiveness due to confounding risk, and expert committee reports (option B) are typically considered lower-level evidence unless they are explicitly based on systematic evidence review methods. For implementing CAUTI prevention changes, relying first on systematic syntheses best supports standardized, evidence-based practice.
During an infection control round in the operating room, the infection preventionist (IP) notices that sterile instrument pouches do not have a sterilization expiration date. What is the MOST appropriate action for the IP to take?
Allow the use of pouches, as long as they appear clean and their integrity is intact.
Instruct staff to discard all sterile instrument pouches without an expiration date immediately.
Require the Sterile Processing Department to apply a standard expiration date of 30 days on all sterile pouches.
Confirm that the facility follows an event-related shelf-life policy and verify the integrity of the pouches and storage conditions.
The CBIC Certified Infection Control Exam Study Guide (6th edition) explains that sterile items are no longer managed using time-related expiration dating but rather by event-related shelf life. Under an event-related shelf-life system, sterile items remain sterile indefinitely unless an event occurs that compromises their integrity, such as package damage, moisture exposure, improper handling, or poor storage conditions.
Therefore, the absence of an expiration date on sterile instrument pouches does not automatically indicate noncompliance or require disposal. The most appropriate action for the infection preventionist is to verify that the facility has a written event-related shelf-life policy and to assess whether sterile packages are intact, properly sealed, clean, dry, and stored under appropriate environmental conditions. This approach aligns with nationally recognized standards and current evidence-based practice.
Option A is incomplete because it does not ensure that a formal policy and appropriate storage practices are in place. Option B is unnecessary and wasteful when no compromise of sterility has occurred. Option C is incorrect because arbitrarily assigning a time-based expiration (e.g., 30 days) contradicts modern sterilization principles and is not evidence-based.
For the CIC® exam, this question reinforces the principle that sterility is event-related, not time-related, and that infection preventionists must evaluate policies, storage conditions, and package integrity rather than defaulting to unnecessary disposal.
In the current year, cases of tuberculosis (TB) among foreign-born persons accounted for the majority of new TB cases in the United States. The number of states with greater than 50% of cases among foreign-born persons increased from four cases ten years ago to 22 cases in the current year. This information can BEST be used to
heighten awareness among Emergency Department staff.
inform staff who are foreign-born.
educate patients and visitors.
review the TB exposure control plan.
1 and 2 only.
1 and 4 only.
2 and 3 only.
3 and 4 only.
The correct answer is B, "1 and 4 only," indicating that the information can best be used to heighten awareness among Emergency Department (ED) staff and review the TB exposure control plan. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, tuberculosis (TB) remains a significant public health concern, particularly with the increasing proportion of cases among foreign-born persons in the United States. The data showing a rise from four to 22 states with over 50% of TB cases among foreign-born individuals highlights an evolving epidemiological trend that warrants targeted infection prevention strategies (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.1 - Conduct surveillance for healthcare-associated infections and epidemiologically significant organisms).
Heightening awareness among ED staff (option 1) is critical because the ED is often the first point of contact for patients with undiagnosed or active TB, especially those from high-prevalence regions. Increased awareness can improve early identification, isolation, and reporting of potential cases. Reviewing the TB exposure control plan (option 4) is equally important, as it allows the infection preventionist to assess and update protocols—such as ventilation, personal protective equipment (PPE) use, and screening processes—to address the heightened risk posed by the growing number of cases among foreign-born individuals (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents).
Option 2 (inform staff who are foreign-born) is not the best use of this data, as the information pertains to patient demographics rather than staff risk, and targeting staff based on their origin could be inappropriate without specific exposure evidence. Option 3 (educate patients and visitors) is a general education strategy but less directly actionable with this specific epidemiological data, which is more relevant to healthcare worker preparedness and facility protocols. Combining options 1 and 4 aligns with CBIC’s emphasis on using surveillance data to guide prevention and control measures, ensuring a proactive response to the increased TB burden (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.5 - Use data to guide infection prevention and control strategies).
An infection preventionist is informed that there is a possible cluster of streptococcal meningitis in the neonatal intensive care unit. Which of the following streptococcal serogroops is MOST commonly associated with meningitis in neonates beyond one week of age?
Group A
Group B
Group C
Group D
Group B Streptococcus (Streptococcus agalactiae) is the most common cause of neonatal bacterial meningitis beyond one week of age.
Step-by-Step Justification:
Group B Streptococcus (GBS) and Neonatal Infections:
GBS is a leading cause of late-onset neonatal meningitis (occurring after 7 days of age).
Infection typically occurs through vertical transmission from the mother or postnatal exposure.
Neonatal Risk Factors:
Premature birth, prolonged rupture of membranes, and maternal GBS colonization increase risk.
Why Other Options Are Incorrect:
A. Group A: Rare in neonates and more commonly associated with pharyngitis and skin infections.
C. Group C: Typically associated with animal infections and rarely affects humans.
D. Group D: Includes Enterococcus, which can cause neonatal infections but is not the most common cause of meningitis.
CBIC Infection Control References:
APIC Text, "Group B Streptococcus and Neonatal Meningitis".
An infection preventionist, Cancer Committee, and Intravenous Therapy Department are studying the incidence of infections in patients with triple lumen catheters. Which of the following is essential to the quality improvement process?
Establish subjective criteria for outcome measurement.
Recommendations for intervention must be approved by the governing board.
Study criteria must be approved monthly by the Cancer Committee.
A monitoring system must be in place following implementation of interventions.
The correct answer is D, "A monitoring system must be in place following implementation of interventions," as this is essential to the quality improvement (QI) process. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, a key component of any QI initiative, such as studying the incidence of infections in patients with triple lumen catheters, is the continuous evaluation of interventions to assess their effectiveness and ensure sustained improvement. A monitoring system allows the infection preventionist (IP), Cancer Committee, and Intravenous Therapy Department to track infection rates, identify trends, and make data-driven adjustments to infection control practices post-intervention (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.4 - Evaluate the effectiveness of infection prevention and control interventions). This step is critical to validate the success of implemented strategies, such as catheter care protocols, and to prevent healthcare-associated infections (HAIs).
Option A (establish subjective criteria for outcome measurement) is not ideal because QI processes rely on objective, measurable outcomes (e.g., infection rates per 1,000 catheter days) rather than subjective criteria to ensure reliability and reproducibility. Option B (recommendations for intervention must be approved by the governing board) is an important step for institutional support and resource allocation, but it is a preparatory action rather than an essential component of the ongoing QI process itself. Option C (study criteria must be approved monthly by the Cancer Committee) suggests an unnecessary administrative burden; while initial approval of study criteria is important, monthly re-approval is not a standard QI requirement unless mandated by specific policies, and it does not directly contribute to the improvement process.
The emphasis on a monitoring system aligns with CBIC’s focus on using surveillance data to guide and refine infection prevention efforts, ensuring that interventions for triple lumen catheter-related infections are effective and adaptable (CBIC Practice Analysis, 2022, Domain II: Surveillance and Epidemiologic Investigation, Competency 2.5 - Use data to guide infection prevention and control strategies). This approach supports a cycle of continuous improvement, which is foundational to reducing catheter-associated bloodstream infections (CABSI) in healthcare settings.
Which of the following activities will BEST prepare a newly hired infection preventionist to present information at the facility’s orientation program?
Observing other departments’ orientation presentations
Meeting with the facility’s leadership
Reviewing principles of adult learning
Administering tuberculin skin tests to orientees
The correct answer is C, "Reviewing principles of adult learning," as this activity will best prepare a newly hired infection preventionist to present information at the facility’s orientation program. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, effective education delivery, especially for healthcare professionals during orientation, relies on understanding adult learning principles (e.g., andragogy), which emphasize learner-centered approaches, relevance to practice, and active participation. Reviewing these principles equips the infection preventionist (IP) to design and deliver content that addresses the specific needs, experiences, and motivations of the audience—such as new staff learning infection control protocols—enhancing engagement and retention (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.1 - Develop and implement educational programs). This preparation ensures the presentation is tailored, impactful, and aligned with the goal of promoting infection prevention behaviors.
Option A (observing other departments’ orientation presentations) can provide insights into presentation styles or facility norms, but it is less focused on the IP’s specific educational role and may not address the unique content of infection prevention. Option B (meeting with the facility’s leadership) is valuable for understanding organizational priorities and gaining support, but it is more about collaboration and context-setting rather than direct preparation for presenting educational material. Option D (administering tuberculin skin tests to orientees) is a clinical task related to TB screening, not a preparatory activity for designing or delivering an educational presentation.
The focus on reviewing adult learning principles aligns with CBIC’s emphasis on evidence-based education strategies to improve infection control practices among healthcare personnel (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.2 - Evaluate the effectiveness of educational programs). This approach enables the IP to effectively communicate critical information, such as hand hygiene or isolation protocols, during the orientation program.
An outbreak of Candida auris is suspected in the infection preventionist's (IP) facility. The IP's investigation must be conducted in a standard method and communication is critical. Which first step is MOST important?
Conduct environmental cultures
Plan to prevent future outbreaks
Notify facility administration
Perform analytical studies
In an outbreak investigation, the first critical step is to notify facility administration and other key stakeholders. This ensures the rapid mobilization of resources, coordination with infection control teams, and compliance with regulatory reporting requirements.
Why the Other Options Are Incorrect?
A. Conduct environmental cultures – While environmental sampling may be necessary, it is not the first step. The outbreak must first be confirmed and administration alerted.
B. Plan to prevent future outbreaks – Prevention planning happens later after the outbreak has been investigated and controlled.
D. Perform analytical studies – Data analysis occurs after case definition and initial response measures are in place.
CBIC Infection Control Reference
APIC guidelines state that the first step in an outbreak investigation is confirming the outbreak and notifying key stakeholders.
Catheter associated urinary tract infection (CAUTI) improvement team is working to decrease CAUTIs in the hospital. Which of the following would be a process measure that would help to reduce CAUTI?
CAUTI rate per 1000 catheter days
Standardized Infection Ratio per unit
Rate of bloodstream infections secondary to CAUTI
Staff compliance to proper insertion technique
A process measure assesses how well healthcare personnel follow specific procedures known to prevent infection. In the case of CAUTI (Catheter-Associated Urinary Tract Infection), monitoring staff compliance with proper insertion technique is a direct process measure.
According to the APIC/JCR Workbook, effective CAUTI prevention involves evaluating compliance with proper catheter insertion and maintenance practices. Monitoring this behavior is a process measure that directly affects outcomes like infection rate reduction.
The CBIC Study Guide also emphasizes using compliance with evidence-based insertion techniques as a strategy to measure and improve CAUTI prevention efforts.
APIC Text notes that “a process measure focuses on a process or the steps in a process that leads to a specific outcome.” This includes monitoring healthcare staff performance related to proper catheter insertion and care.
Incorrect answer rationale:
A. CAUTI rate per 1000 catheter days – This is an outcome measure, not a process measure.
B. Standardized Infection Ratio per unit – Also an outcome/benchmarking metric.
C. Rate of bloodstream infections secondary to CAUTI – This is an outcome, not a process.
Which of the following reasons BEST describes the importance of documenting cleaning, disinfection, and sterilization processes?
Reduce the cost of hospital operations.
Ensure compliance with Spaulding classification scheme.
Ensure that all processes are conducted on a regular basis.
Comply with policies, regulations, and accreditation standards.
The Certification Study Guide (6th edition) emphasizes that documentation of cleaning, disinfection, and sterilization processes is a fundamental requirement for regulatory compliance and patient safety assurance. Accurate and complete documentation demonstrates that reprocessing activities are performed according to established policies, manufacturer instructions for use (IFUs), and evidence-based standards. This documentation is essential for meeting expectations set by regulatory agencies, accrediting bodies, and internal quality assurance programs.
Documentation provides verifiable proof that critical steps—such as cleaning, monitoring of sterilization parameters, load release, and equipment maintenance—have been performed correctly. In the event of a healthcare-associated infection investigation, recall, or survey, records serve as objective evidence that proper reprocessing practices were followed. The study guide highlights that “if it is not documented, it is considered not done”, a principle commonly tested on the CIC exam.
The other options reflect secondary or indirect benefits but do not represent the primary reason for documentation. Cost reduction is not the intent of reprocessing records. While Spaulding classification informs how items should be reprocessed, documentation alone does not ensure compliance with that framework. Ensuring processes occur regularly is an operational issue rather than a documentation purpose.
CIC exam questions frequently reinforce that documentation supports accountability, traceability, regulatory compliance, and accreditation readiness, making compliance with policies, regulations, and standards the best answer.
The infection preventionist (IP) is working with the Product Evaluation Committee to select a sporicidal disinfectant for Clostridioides difficile. An effective disinfectant for the IP to recommend is
quaternary ammonium compound.
phenolic.
isopropyl alcohol.
sodium hypochlorite.
The correct answer is D, "sodium hypochlorite," as it is an effective sporicidal disinfectant for Clostridioides difficile that the infection preventionist (IP) should recommend. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, Clostridioides difficile (C. difficile) is a spore-forming bacterium responsible for significant healthcare-associated infections (HAIs), and its spores are highly resistant to many common disinfectants. Sodium hypochlorite (bleach) is recognized by the Centers for Disease Control and Prevention (CDC) and the Environmental Protection Agency (EPA) as a sporicidal agent capable of inactivating C. difficile spores when used at appropriate concentrations (e.g., 1:10 dilution of household bleach) and with the recommended contact time (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). This makes it a preferred choice for environmental disinfection in outbreak settings or areas with known C. difficile contamination.
Option A (quaternary ammonium compound) is effective against many bacteria and viruses but lacks sufficient sporicidal activity against C. difficile spores, rendering it inadequate for this purpose. Option B (phenolic) has broad-spectrum antimicrobial properties but is not reliably sporicidal and is less effective against C. difficile spores compared to sodium hypochlorite. Option C (isopropyl alcohol) is useful for disinfecting surfaces and killing some pathogens, but it is not sporicidal and evaporates quickly, making it ineffective against C. difficile spores.
The IP’s recommendation of sodium hypochlorite aligns with CBIC’s emphasis on selecting disinfectants based on their efficacy against specific pathogens and adherence to evidence-based guidelines (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). Proper use, including correct dilution and contact time, is critical to ensure effectiveness, and the IP should collaborate with the Product Evaluation Committee to ensure implementation aligns with safety and regulatory standards (CDC Guidelines for Environmental Infection Control in Healthcare Facilities, 2019).
Which of the following individuals should be excluded from receiving live attenuated influenza virus?
Pregnant persons
Healthy persons aged 2 to 49
Persons with allergies to chicken feathers
Persons simultaneously receiving an inactivated vaccine
The correct answer is A, "Pregnant persons," as they should be excluded from receiving the live attenuated influenza virus (LAIV) vaccine. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which align with recommendations from the Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP), the LAIV, commonly known as the nasal spray flu vaccine, contains a live attenuated form of the influenza virus. This vaccine is contraindicated in pregnant individuals due to the theoretical risk of the attenuated virus replicating and potentially harming the fetus, despite limited evidence of adverse outcomes (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). Pregnant persons are instead recommended to receive the inactivated influenza vaccine (IIV), which is considered safe during pregnancy.
Option B (healthy persons aged 2 to 49) is incorrect because this group is generally eligible to receive LAIV, provided they have no other contraindications, as the vaccine is approved for healthy, non-pregnant individuals in this age range (CDC Immunization Schedules, 2024). Option C (persons with allergies to chicken feathers) is not a contraindication for LAIV; the vaccine is produced in eggs, and while egg allergy was historically a concern, current guidelines indicate that LAIV can be administered to persons with egg allergies if they can tolerate egg in their diet, with precautions managed by healthcare providers. Option D (persons simultaneously receiving an inactivated vaccine) is also incorrect, as LAIV can be co-administered with inactivated vaccines without issue, according to ACIP recommendations, as there is no significant interference between the two vaccine types.
The exclusion of pregnant persons reflects CBIC’s emphasis on tailoring infection prevention strategies, including vaccination programs, to protect vulnerable populations while minimizing risks (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This decision is based on precautionary principles outlined in CDC and ACIP guidelines to ensure maternal and fetal safety (CDC Prevention and Control of Seasonal Influenza with Vaccines, 2023).
Which of the following is the BEST study design for assessing the benefit of a new treatment?
Interrupted time series
Correlational study
Parallel group study
Randomized controlled trial
The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies the randomized controlled trial (RCT) as the gold standard study design for assessing the benefit of a new treatment. RCTs are specifically designed to determine causality by minimizing bias and confounding variables through random assignment of participants to intervention and control groups. This ensures that differences in outcomes can be attributed with the highest level of confidence to the treatment being studied rather than to external factors.
In an RCT, participants are randomly allocated to receive either the new treatment or a comparison intervention (such as standard therapy or placebo). Randomization balances known and unknown risk factors between groups, while controlled conditions allow precise measurement of treatment effects. This design is particularly important when evaluating new therapies, medications, or interventions where efficacy and safety must be clearly demonstrated.
The other study designs listed are less rigorous for assessing treatment benefit. An interrupted time series is useful for evaluating system-level interventions over time but is more susceptible to confounding influences. A correlational study can identify associations but cannot establish cause and effect. A parallel group study without randomization lacks adequate control for bias and confounding.
For CIC® exam preparation, it is essential to recognize that when the objective is to assess the benefit or effectiveness of a new treatment, a randomized controlled trial provides the strongest and most reliable evidence, making it the best answer.
The infection preventionist recognizes that facility renovation projects are associated with risk for healthcare-associated infections (HAIs). What is the primary rationale for this risk?
Increased numbers of construction staff resulting in congested workspaces
Improper training regarding hand hygiene for contracted workers
Trash removal using uncovered carts
Environmental dispersal of microorganisms
The Certification Study Guide (6th edition) identifies environmental dispersal of microorganisms as the primary reason healthcare construction and renovation activities increase the risk of healthcare-associated infections (HAIs). Construction activities such as demolition, drilling, and ceiling penetration disturb dust and building materials that may harbor fungal spores and other microorganisms, particularly Aspergillus species. Once aerosolized, these organisms can spread through airflow to patient care areas if containment and ventilation controls are inadequate.
The study guide emphasizes that immunocompromised patients—such as those in oncology units, transplant units, and intensive care settings—are especially vulnerable to infections caused by airborne fungi released during construction. Numerous outbreaks of invasive aspergillosis have been linked directly to renovation and construction projects that lacked appropriate infection control risk assessment (ICRA) measures.
The incorrect options describe secondary or contributory issues but are not the primary mechanism of infection risk. While increased personnel traffic, hand hygiene training, and waste handling are important considerations, they do not represent the central hazard posed by construction. The fundamental risk is airborne dissemination of microorganisms from disrupted environmental reservoirs.
CIC exam questions frequently test knowledge of construction-related infection risks and the importance of engineering controls such as negative pressure containment, HEPA filtration, and dust barriers. Recognizing environmental dispersal as the primary risk underscores why rigorous planning and infection control oversight are essential during renovation projects.
An infection preventionist is notified of a patient with Gram negative diplococci from a cerebral spinal fluid specimen. The patient was intubated during ambulance transport and intravenous lines are placed after arrival to the Emergency Department (ED). The patient was immediately placed in Droplet Precautions upon admission to the ED. Which of the following statements is true regarding the need for evaluating exposure to communicable illness?
Follow-up evaluation is not required for this laboratory finding.
ED personnel should be evaluated for possible exposure.
Ambulance personnel should be evaluated for possible exposure.
Follow-up evaluation is not necessary as the appropriate precautions were promptly instituted.
The correct answer is C, "Ambulance personnel should be evaluated for possible exposure," as this statement is true regarding the need for evaluating exposure to communicable illness. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, the presence of Gram negative diplococci in a cerebral spinal fluid (CSF) specimen is suggestive of a serious bacterial infection, most likely Neisseria meningitidis, which causes meningococcal disease. This condition is highly contagious and can be transmitted through respiratory droplets or direct contact with respiratory secretions, particularly during procedures like intubation (CBIC Practice Analysis, 2022, Domain I: Identification of Infectious Disease Processes, Competency 1.1 - Identify infectious disease processes). The patient was intubated during ambulance transport, creating a potential aerosol-generating procedure (AGP) that could have exposed ambulance personnel to infectious droplets before Droplet Precautions were instituted upon arrival at the Emergency Department (ED). Therefore, evaluating ambulance personnel for possible exposure is necessary to assess their risk and determine if post-exposure prophylaxis (e.g., antibiotics) or monitoring is required.
Option A (follow-up evaluation is not required for this laboratory finding) is incorrect because the identification of Gram negative diplococci in CSF is a critical finding that warrants investigation due to the potential for meningococcal disease, a reportable and transmissible condition. Option B (ED personnel should be evaluated for possible exposure) is less applicable since the patient was immediately placed in Droplet Precautions upon ED admission, minimizing exposure risk to ED staff after that point, though it could be considered if exposure occurred before precautions were fully implemented. Option D (follow-up evaluation is not necessary as the appropriate precautions were promptly instituted) is inaccurate because the prompt institution of Droplet Precautions in the ED does not retroactively address the exposure risk during ambulance transport, where precautions were not in place.
The focus on evaluating ambulance personnel aligns with CBIC’s emphasis on identifying and mitigating transmission risks associated with communicable diseases, particularly in high-risk settings like ambulance transport (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). This step is supported by CDC guidelines, which recommend exposure evaluation and prophylaxis for close contacts of meningococcal disease cases (CDC Meningococcal Disease Management, 2021).
What is the MOST effective way an infection preventionist can assess readiness of emergency preparedness plans for an influx of patients with an emerging viral hemorrhagic fever?
Meet frequently with emergency management professionals in the hospital and local public health authority.
Conduct regular rounding in the Emergency Department providing education and reviewing policies and procedures with frontline staff
Coordinate with hospital-based emergency management professionals and other incident command stakeholders to conduct a tabletop exercise or full-scale drill.
Collaborate with hospital stakeholders to assess the current availability of backup supplies of both staff and personal protective equipment
The most effective way to assess emergency preparedness for an influx of patients with viral hemorrhagic fever (VHF) is through tabletop exercises or full-scale drills. These exercises simulate real-life scenarios, allowing hospitals to test protocols, identify weaknesses, and improve response efforts.
Why the Other Options Are Incorrect?
A. Meet frequently with emergency management professionals – While important, meetings alone do not provide hands-on testing of preparedness.
B. Conduct regular rounding in the Emergency Department – Rounding helps with policy compliance, but does not test the entire emergency response plan.
D. Collaborate to assess the availability of supplies and PPE – This is one component of preparedness but does not evaluate the facility’s response in real-time.
CBIC Infection Control Reference
APIC recommends full-scale emergency drills as the gold standard for assessing preparedness for emerging infectious diseases.
An infection preventionist is writing a policy about prevention of intravascular device infection. Which of the following is important for healthcare personnel to know as part of central line insertion and maintenance procedures?
Change the central line every seven days.
Use maximum sterile barrier precautions for the line insertion.
The femoral site is the preferred site of insertion in an adult patient.
Use 70% isopropyl alcohol for skin preparation before line insertion.
The Certification Study Guide (6th edition) identifies the use of maximum sterile barrier (MSB) precautions during central line insertion as a cornerstone practice for preventing intravascular device–associated infections, including central line–associated bloodstream infections (CLABSIs). MSB precautions include wearing a cap, mask, sterile gown, and sterile gloves, and using a large sterile drape to fully cover the patient during line insertion. These measures significantly reduce the risk of introducing skin flora and environmental microorganisms into the bloodstream at the time of catheter placement.
The study guide emphasizes that the highest risk for contamination occurs during insertion, making strict aseptic technique essential. MSB precautions are a required element of evidence-based central line insertion bundles and are consistently associated with reduced CLABSI rates when reliably implemented.
The other options reflect outdated or incorrect practices. Routine scheduled replacement of central lines every seven days is not recommended and does not reduce infection risk. The femoral vein is not the preferred insertion site in adults due to higher infection risk compared to subclavian or internal jugular sites. While alcohol is used during hub disinfection, chlorhexidine-based antisepsis (preferably chlorhexidine with alcohol) is recommended for skin preparation—not alcohol alone.
This question highlights a core CIC exam concept: standardized insertion practices using maximum sterile barriers are among the most effective strategies for preventing intravascular device infections.
The effectiveness of disinfection is NOT dependent upon the:
Virulence of the organism
Amount of organic material present
Spore-forming ability of the organism
Length of exposure to the chemical agent
The CBIC Certified Infection Control Exam Study Guide (6th edition) explains that the effectiveness of disinfection depends on multiple physical, chemical, and biologic factors, but virulence of the organism is not one of them. Virulence refers to an organism’s ability to cause disease in a host, which is a clinical characteristic, not a determinant of susceptibility to disinfectants.
Disinfection efficacy is influenced by factors such as the type and number of microorganisms, particularly their intrinsic resistance (for example, spores are more resistant than vegetative bacteria), making option C a true determinant. The amount of organic material present (option B) is also critical, as organic matter can inactivate disinfectants or shield microorganisms from exposure. Likewise, the length of exposure (contact time) to the chemical agent (option D) is essential to achieving the desired level of microbial kill and is specified in manufacturer instructions for use.
Virulence does not affect how easily an organism is destroyed by a disinfectant. For example, a highly virulent organism may be easily killed by a low-level disinfectant, while a less virulent organism such as a bacterial spore may be highly resistant. Therefore, virulence plays no role in determining disinfection effectiveness.
For CIC® exam preparation, it is important to distinguish between clinical severity and microbial resistance. Disinfection effectiveness is based on resistance characteristics and process variables—not on how dangerous the organism is to humans.
A new hospital disinfectant with a 3-minute contact time has been purchased by Environmental Services. The disinfectant will be rolled out across the patient care 3-minute contact time has been purchased by Environmental Services. The disinfectant will be rolled out across the patient care areas. They are concerned about the high cost of the disinfectant. What advice can the infection preventionist provide?
Use the new disinfectant for patient washrooms only.
Use detergents on the floors in patient rooms.
Use detergents on smooth horizontal surfaces.
Use new disinfectant for all surfaces in the patient room.
The scenario involves the introduction of a new hospital disinfectant with a 3-minute contact time, intended for use across patient care areas, but with concerns raised by Environmental Services about its high cost. The infection preventionist’s advice must balance infection control efficacy with cost management, adhering to principles outlined by the Certification Board of Infection Control and Epidemiology (CBIC) and evidence-based practices. The goal is to optimize the disinfectant’s use while ensuring a safe environment. Let’s evaluate each option:
A. Use the new disinfectant for patient washrooms only: Limiting the disinfectant to patient washrooms focuses its use on high-touch, high-risk areas where pathogens (e.g., Clostridioides difficile, norovirus) may be prevalent. However, this approach restricts the disinfectant’s application to a specific area, potentially leaving other patient care surfaces (e.g., bed rails, tables) vulnerable to contamination. While cost-saving, it does not address the broad infection control needs across all patient care areas, making it an incomplete strategy.
B. Use detergents on the floors in patient rooms: Detergents are cleaning agents that remove dirt and organic material but lack the antimicrobial properties of disinfectants. Floors in patient rooms can harbor pathogens, but they are generally considered lower-risk surfaces compared to high-touch areas (e.g., bed rails, doorknobs). Using detergents instead of the new disinfectant on floors could reduce costs but compromises infection control, as floors may still contribute to environmental transmission (e.g., via shoes or equipment). This option is not optimal given the availability of an effective disinfectant.
C. Use detergents on smooth horizontal surfaces: Smooth horizontal surfaces (e.g., tables, counters, overbed tables) are common sites for pathogen accumulation and transmission in patient rooms. Using detergents to clean these surfaces removes organic material, which is a critical first step before disinfection. If the 3-minute contact time disinfectant is reserved for high-touch or high-risk surfaces (e.g., bed rails, call buttons) where disinfection is most critical, this approach maximizes the disinfectant’s efficacy while reducing its overall use and cost. This strategy aligns with CBIC guidelines, which emphasize a two-step process (cleaning followed by disinfection) and targeted use of resources, making it a practical and cost-effective recommendation.
D. Use new disinfectant for all surfaces in the patient room: Using the disinfectant on all surfaces ensures comprehensive pathogen reduction but increases consumption and cost, which is a concern for Environmental Services. While the 3-minute contact time suggests efficiency, overusing the disinfectant on low-risk surfaces (e.g., floors, walls) may not provide proportional infection control benefits and could strain the budget. This approach does not address the cost concern and is less strategic than targeting high-risk areas.
The best advice is C, using detergents on smooth horizontal surfaces to handle routine cleaning, while reserving the new disinfectant for high-touch or high-risk areas where its antimicrobial action is most needed. This optimizes infection prevention, aligns with CBIC’s emphasis on evidence-based environmental cleaning, and addresses the cost concern by reducing unnecessary disinfectant use. The infection preventionist should also recommend a risk assessment to identify priority surfaces for disinfectant application.
CBIC Infection Prevention and Control (IPC) Core Competency Model (updated 2023), Domain IV: Environment of Care, which advocates for targeted cleaning and disinfection based on risk.
CBIC Examination Content Outline, Domain III: Prevention and Control of Infectious Diseases, which includes cost-effective use of disinfectants.
CDC Guidelines for Environmental Infection Control in Healthcare Facilities (2022), which recommend cleaning with detergents followed by targeted disinfection.
What is the correct order of steps for reprocessing critical medical equipment?
Clean, sterilize, disinfect
Disinfect, clean, sterilize
Disinfect, sterilize
Clean, sterilize
The correct answer is D, "Clean, sterilize," as this represents the correct order of steps for reprocessing critical medical equipment. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, critical medical equipment—items that enter sterile tissues or the vascular system (e.g., surgical instruments, implants)—must undergo a rigorous reprocessing cycle to ensure they are free of all microorganisms, including spores. The process begins with cleaning to remove organic material, debris, and soil, which is essential to allow subsequent sterilization to be effective. Sterilization, the final step, uses methods such as steam, ethylene oxide, or hydrogen peroxide gas to achieve a sterility assurance level (SAL) of 10⁻⁶, eliminating all microbial life (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). Disinfection, while important for semi-critical devices, is not a step in the reprocessing of critical items, as it does not achieve the sterility required; it is a separate process for non-critical or semi-critical equipment.
Option A (clean, sterilize, disinfect) is incorrect because disinfecting after sterilization is unnecessary and redundant, as sterilization already achieves a higher level of microbial kill. Option B (disinfect, clean, sterilize) reverses the logical sequence; cleaning must precede any disinfection or sterilization to remove bioburden, and disinfection is not appropriate for critical items. Option C (disinfect, sterilize) omits cleaning and incorrectly prioritizes disinfection, which is insufficient for critical equipment requiring full sterility.
The focus on cleaning followed by sterilization aligns with CBIC’s emphasis on evidence-based reprocessing protocols to prevent healthcare-associated infections (HAIs), ensuring that critical equipment is safe for patient use (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). This sequence is supported by standards such as AAMI ST79, which outlines the mandatory cleaning step before sterilization to ensure efficacy and safety.
An HBsAb-negative employee has a percutaneous exposure to blood from a Hepatitis B surface antigen (HBsAg) positive patient. Which of the following regimens is recommended for this employee?
Immune serum globulin and hepatitis B vaccine
Hepatitis B immune globulin (HBIG) alone
Hepatitis B vaccine alone
Hepatitis B immune globulin (HBIG) and hepatitis B vaccine
The correct answer is D, "Hepatitis B immune globulin (HBIG) and hepatitis B vaccine," as this is the recommended regimen for an HBsAb-negative employee with a percutaneous exposure to blood from an HBsAg-positive patient. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which align with recommendations from the Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP), post-exposure prophylaxis (PEP) for hepatitis B virus (HBV) exposure depends on the employee’s vaccination status and the source’s HBsAg status. For an unvaccinated or known HBsAb-negative individual (indicating no immunity) exposed to HBsAg-positive blood, the standard PEP includes both HBIG and the hepatitis B vaccine. HBIG provides immediate passive immunity by delivering pre-formed antibodies, while the vaccine initiates active immunity to prevent future infections (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.2 - Implement measures to prevent transmission of infectious agents). The HBIG should be administered within 24 hours of exposure (preferably within 7 days), and the first dose of the vaccine should be given concurrently, followed by the complete vaccine series.
Option A (immune serum globulin and hepatitis B vaccine) is incorrect because immune serum globulin (ISG) is a general immunoglobulin preparation and not specific for HBV; HBIG, which contains high titers of anti-HBs, is the appropriate specific immunoglobulin for HBV exposure. Option B (hepatitis B immune globulin [HBIG] alone) is insufficient, as it provides only temporary passive immunity without initiating long-term active immunity through vaccination, which is critical for an unvaccinated individual. Option C (hepatitis B vaccine alone) is inadequate for immediate post-exposure protection, as it takes weeks to develop immunity, leaving the employee vulnerable in the interim.
The recommendation for HBIG and hepatitis B vaccine aligns with CBIC’s emphasis on evidence-based post-exposure management to prevent HBV transmission in healthcare settings (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This dual approach is supported by CDC guidelines, which prioritize rapid intervention to reduce the risk of seroconversion following percutaneous exposure (CDC Updated U.S. Public Health Service Guidelines for the Management of Occupational Exposures to HBV, HCV, and HIV, 2013).
A hospital wants to launch an alternative care site due to an influx of patients from a mass casualty incident. An infection preventionist should be engaged to BEST determine:
Optimal medical care being offered.
Staffing of licensed and unlicensed staff.
Measures to keep all individuals healthy.
Requirements for providing direct patient care.
The Certification Study Guide (6th edition) outlines the critical role of the infection preventionist (IP) in emergency preparedness and response, particularly when healthcare systems activate alternate or alternative care sites during mass casualty incidents or public health emergencies. In these situations, the IP’s primary responsibility is to determine the infection prevention and control requirements necessary to safely provide direct patient care in nontraditional settings.
Alternate care sites often lack the infrastructure of acute care hospitals, such as standard ventilation, hand hygiene facilities, isolation rooms, or routine environmental services. The study guide emphasizes that infection preventionists must assess risks related to patient placement, cohorting, isolation precautions, environmental cleaning, waste management, water safety, and availability of personal protective equipment. These determinations directly influence whether patient care can be delivered safely and sustainably under emergency conditions.
The other options fall outside the IP’s primary scope. Decisions about optimal medical care and staffing models are led by clinical and administrative leadership. “Measures to keep all individuals healthy” is overly broad and does not reflect the IP’s focused, operational role during emergency site activation.
CIC exam questions frequently test understanding of role delineation during emergency management. The infection preventionist’s expertise is best applied to defining infection control standards and requirements that enable safe direct patient care—making option D the most accurate and appropriate answer.
During a COVID outbreak with hospital-associated transmission cases, the infection preventionist (IP) receives a news media call about what is being done to reduce the transmission. The IP's BEST response is to
answer the questions truthfully.
give vague answers to ensure patient privacy.
refer the reporters to the hospital's media spokesperson.
inform the reporter that the conversation must be recorded to ensure accuracy.
The best response for an infection preventionist (IP) when receiving a news media call during a COVID outbreak with hospital-associated transmission cases is to refer the reporters to the hospital's media spokesperson. This approach aligns with the principles outlined in the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, which emphasize the importance of maintaining professionalism, protecting patient privacy, and ensuring accurate communication. The IP's primary role is to focus on infection prevention and control activities rather than serving as a public relations representative. Engaging directly with the media can risk divulging sensitive patient information or operational details that may not be fully contextualized, potentially violating the Health Insurance Portability and Accountability Act (HIPAA) or other privacy regulations.
Option A (answer the questions truthfully) is not ideal because, while truthfulness is important, the IP may not have the authority or full context to provide a comprehensive and accurate public statement, and doing so could inadvertently compromise patient confidentiality or misrepresent hospital policies. Option B (give vague answers to ensure patient privacy) might protect privacy but could lead to miscommunication or lack of trust if the responses appear evasive without a clear referral process. Option D (inform the reporter that the conversation must be recorded to ensure accuracy) is a procedural step but does not address the core issue of who should handle media inquiries.
Referring to the hospital's media spokesperson (Option C) ensures that a trained individual handles the communication, adhering to CBIC's emphasis on collaboration with organizational leadership and adherence to institutional communication protocols (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.1 - Collaborate with organizational leaders). This also aligns with best practices for managing public health crises, where centralized and coordinated messaging is critical to avoid misinformation.
Which of the following is an essential element of practice when sending biohazardous samples from one location to another?
Ship using triple-containment packaging
Electronically log and send via overnight delivery
Transport by an authorized biohazard transporter
Store in a cooler that is labeled as a health hazard
The safe transport of biohazardous samples, such as infectious agents, clinical specimens, or diagnostic materials, is a critical aspect of infection prevention and control to prevent exposure and environmental contamination. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes adherence to regulatory and safety standards in the "Prevention and Control of Infectious Diseases" domain, which includes proper handling and shipping of biohazardous materials. The primary guideline governing this practice is the U.S. Department of Transportation (DOT) Hazardous Materials Regulations (HMR) and the International Air Transport Association (IATA) Dangerous Goods Regulations, which align with global biosafety standards.
Option A, "Ship using triple-containment packaging," is the essential element of practice. Triple-containment packaging involves three layers: a primary watertight container holding the sample, a secondary leak-proof container with absorbent material, and an outer rigid packaging (e.g., a box) that meets shipping regulations. This system ensures that biohazardous materials remain secure during transport, preventing leaks or breaches that could expose handlers or the public. The CDC and WHO endorse this method as a fundamental requirement for shipping Category A (high-risk) and Category B (moderate-risk) infectious substances, making it the cornerstone of safe transport practice.
Option B, "Electronically log and send via overnight delivery," is a useful administrative and logistical step to track shipments and ensure timely delivery, but it is not the essential element. While documentation and rapid delivery are important for maintaining chain of custody and sample integrity, they are secondary to the physical containment provided by triple packaging. Option C, "Transport by an authorized biohazard transporter," is a necessary step to comply with regulations, as only trained and certified transporters can handle biohazardous materials. However, this is contingent on proper packaging; without triple containment, transport authorization alone is insufficient. Option D, "Store in a cooler that is labeled as a health hazard," may be part of preparation (e.g., maintaining sample temperature), but labeling alone does not address the containment or transport safety required during shipment. Coolers are often used, but the focus on labeling as a health hazard is incomplete without the triple-containment structure.
The CBIC Practice Analysis (2022) supports compliance with federal and international shipping regulations, which prioritize triple-containment packaging as the foundational practice to mitigate risks. The CDC’s Biosafety in Microbiological and Biomedical Laboratories (BMBL, 6th Edition, 2020) and IATA guidelines further specify that triple packaging is mandatory for all biohazardous shipments, reinforcing Option A as the correct answer.
At a facility with 10.000 employees. 5,000 are at risk for bloodbome pathogen exposure. Over the past five years, 100 of the 250 needlestick injuries involved exposure to bloodborne pathogens, and 2% of exposed employees seroconverted. How many employees became infected?
1
2
5
10
To determine the number of employees who seroconverted (became infected) after a needlestick exposure, we use the given data:
Total Needlestick Injuries: 250
Needlestick Injuries Involving Bloodborne Pathogens: 100
Seroconversion Rate: 2%
Calculation:
Why Other Options Are Incorrect:
A. 1: Incorrect calculation; 2% of 100 is 2, not 1.
C. 5: Overestimates the actual number of infections.
D. 10: Exceeds the calculated value based on given data.
CBIC Infection Control References:
APIC Text, "Occupational Exposure and Seroconversion Risks".
APIC Text, "Bloodborne Pathogens and Needlestick Injury Prevention"
An infection preventionist (IP) is tasked with developing an infection prevention training program for family members. What step should the IP take FIRST?
Assess the needs of the family members at the facility.
Create clearly defined goals and objectives for the training.
Ensure that all content in the training is relevant and practical.
Develop a plan to create an appropriate training environment.
The correct answer is A, "Assess the needs of the family members at the facility," as this is the first step the infection preventionist (IP) should take when developing an infection prevention training program for family members. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, effective education programs begin with a needs assessment to identify the specific knowledge gaps, cultural factors, and practical challenges of the target audience—in this case, family members. This initial step ensures that the training is tailored to their level of understanding, language preferences, and the infection risks they may encounter (e.g., hand hygiene, isolation protocols), aligning with adult learning principles (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.1 - Develop and implement educational programs). Without this assessment, subsequent steps risk being misaligned with the audience’s needs, reducing the program’s effectiveness.
Option B (create clearly defined goals and objectives for the training) is a critical step but follows the needs assessment, as goals should be based on identified needs to ensure relevance. Option C (ensure that all content in the training is relevant and practical) depends on understanding the audience’s needs first, making it a later step in the development process. Option D (develop a plan to create an appropriate training environment) is important for implementation but requires prior knowledge of the audience and content to design effectively.
The focus on assessing needs aligns with CBIC’s emphasis on evidence-based education design, enabling the IP to address specific infection prevention priorities for family members and improve outcomes in the facility (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.2 - Evaluate the effectiveness of educational programs). This approach is supported by CDC guidelines, which recommend audience assessment as a foundational step in health education programs.
Which of the following products or methods is effective for sterilization of heat-sensitive critical items?
Phenolics
Chlorine-based
Quaternary ammonium
Hydrogen peroxide gas plasma
The CBIC Certified Infection Control Exam Study Guide (6th edition) explains that critical items—those that enter sterile tissue or the vascular system—must be sterile at the time of use. When these items are heat-sensitive and cannot tolerate steam sterilization, low-temperature sterilization technologies are required. Among the options listed, hydrogen peroxide gas plasma is an FDA-cleared, low-temperature sterilization method specifically designed for heat- and moisture-sensitive medical devices.
Hydrogen peroxide gas plasma sterilization achieves sterilization by generating reactive free radicals that destroy microorganisms, including bacteria, viruses, fungi, and spores. The study guide emphasizes that this method provides true sterilization rather than disinfection and is widely used for delicate instruments such as certain endoscopes, optical devices, and electronic equipment. It also offers advantages such as short cycle times and minimal toxic residues.
The other options are incorrect because they do not achieve sterilization. Phenolics, chlorine-based products, and quaternary ammonium compounds are disinfectants, not sterilants, and are inappropriate for critical items. Even at high concentrations, these agents cannot reliably destroy bacterial spores and therefore do not meet the definition of sterilization.
This question highlights a key CIC exam concept: critical items require sterilization, and when heat cannot be used, approved low-temperature sterilization technologies such as hydrogen peroxide gas plasma are required to ensure patient safety.
A 2-yoar-old girl is admitted with a fractured tibia. At birth, she was diagnosed with congenital cytomegalovirus (CMV). Which of the following barrier precautions is appropriate for healthcare personnel caring for her?
Wear masks and gloves
Wear gloves when handling body fluids
No barrier precautions are needed
Use gowns, masks, gloves, and a private room
Standard Precautions are sufficient for congenital cytomegalovirus (CMV), which means that gloves should be used when handling body fluids. CMV is primarily transmitted via direct contact with saliva, urine, or blood.
Why the Other Options Are Incorrect?
A. Wear masks and gloves – Masks are not necessary unless performing high-risk aerosol-generating procedures.
C. No barrier precautions are needed – Gloves are required when handling bodily fluids to prevent transmission.
D. Use gowns, masks, gloves, and a private room – CMV does not require Contact or Airborne Precautions.
CBIC Infection Control Reference
APIC guidelines state that CMV transmission is prevented using Standard Precautions, primarily with glove use for body fluid contact.
The BEST roommate selection for a patient with active shingles would be a patient who has had
varicella vaccine.
treatment with acyclovir
a history of herpes simplex.
varicclla zoster immunoglobulin
A patient with active shingles (herpes zoster) is contagious to individuals who have never had varicella (chickenpox) or the varicella vaccine. The best roommate selection is someone who has received the varicella vaccine, as they are considered immune and not at risk for contracting the virus.
Why the Other Options Are Incorrect?
B. Treatment with acyclovir – Acyclovir treats herpes zoster but does not prevent transmission to others.
C. A history of herpes simplex – Prior herpes simplex virus (HSV) infection does not confer immunity to varicella-zoster virus (VZV).
D. Varicella zoster immunoglobulin (VZIG) – VZIG provides temporary immunity but does not offer long-term protection like the vaccine.
CBIC Infection Control Reference
APIC guidelines recommend placing patients with active shingles in a room with individuals immune to varicella, such as those vaccinated.
On January 31, the nursing staff of a long-term care facility reports that five out of 35 residents have developed high fever, nasal discharge, and a dry cough. The BEST diagnostic tool to determine the causative agent is:
Blood culture
Sputum culture
Nasopharyngeal swab
Legionella serology
The scenario describes a cluster of five out of 35 residents in a long-term care facility developing high fever, nasal discharge, and a dry cough, suggesting a potential respiratory infection outbreak. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes the "Identification of Infectious Disease Processes" and "Surveillance and Epidemiologic Investigation" domains, which require selecting the most appropriate diagnostic tool to identify the causative agent promptly. The Centers for Disease Control and Prevention (CDC) provides guidance on diagnostic approaches for respiratory infections, particularly in congregate settings like long-term care facilities.
Option C, "Nasopharyngeal swab," is the best diagnostic tool in this context. The symptoms—high fever, nasal discharge, and a dry cough—are characteristic of upper respiratory infections, such as influenza, respiratory syncytial virus (RSV), or other viral pathogens common in congregate settings. A nasopharyngeal swab is the gold standard for detecting these agents, as it collects samples from the nasopharynx, where many respiratory viruses replicate. The CDC recommends nasopharyngeal swabs for molecular testing (e.g., PCR) to identify viruses like influenza, RSV, or SARS-CoV-2, especially during outbreak investigations in healthcare facilities. The dry cough and nasal discharge align with upper respiratory involvement, making this sample type more targeted than alternatives. Given the potential for rapid spread among vulnerable residents, early identification via nasopharyngeal swab is critical to guide infection control measures.
Option A, "Blood culture," is less appropriate as the best initial tool. Blood cultures are used to detect systemic bacterial infections (e.g., bacteremia or sepsis), but the symptoms described are more suggestive of a primary respiratory infection rather than a bloodstream infection. While secondary bacteremia could occur, blood cultures are not the first-line diagnostic for this presentation and are more relevant if systemic signs (e.g., hypotension) worsen. Option B, "Sputum culture," is useful for lower respiratory infections, such as pneumonia, where productive cough and sputum production are prominent. However, the dry cough and nasal discharge indicate an upper respiratory focus, and sputum may be difficult to obtain from elderly residents, reducing its utility here. Option D, "Legionella serology," is specific for diagnosing Legionella pneumophila, which causes Legionnaires’ disease, typically presenting with fever, cough, and sometimes gastrointestinal symptoms, often in association with water sources. While possible, the lack of mention of pneumonia or water exposure, combined with the upper respiratory symptoms, makes Legionella serology less likely as the best initial test. Serology also requires time for antibody development, delaying diagnosis compared to direct sampling.
The CBIC Practice Analysis (2022) and CDC guidelines for outbreak management in long-term care facilities (e.g., "Prevention Strategies for Seasonal Influenza in Healthcare Settings," 2018) prioritize rapid respiratory pathogen identification, with nasopharyngeal swabs being the preferred method for viral detection. Given the symptom profile and outbreak context, Option C is the most effective and immediate diagnostic tool to determine the causative agent.
Based on the Spaulding classification, which of the following pairings is an example of a semi-critical item and its minimal level of disinfection?
Bedside table; high-level disinfection
Surgical instrument; sterilization
Endocavity probe; high-level disinfection
Bedpan; intermediate-level disinfection
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes the importance of applying Spaulding’s classification to determine the appropriate minimum level of processing for medical devices. Under this system, devices are categorized as critical, semi-critical, or noncritical based on the degree of infection risk associated with their use.
Semi-critical items are those that come into contact with mucous membranes or non-intact skin but do not ordinarily penetrate sterile tissue. Examples include endocavity probes, such as transvaginal or transrectal ultrasound probes. Because mucous membranes are more susceptible to infection than intact skin, semi-critical items require at least high-level disinfection after thorough cleaning to eliminate all microorganisms except large numbers of bacterial spores.
Option C correctly pairs an endocavity probe with high-level disinfection, which is the minimum acceptable level of processing for this classification. Option A is incorrect because a bedside table is a noncritical item and requires only low-level disinfection. Option B describes a critical item, which correctly requires sterilization but does not meet the question’s focus on semi-critical devices. Option D is incorrect because bedpans are noncritical items, and intermediate-level disinfection exceeds the minimum requirement.
Understanding Spaulding’s classification and matching devices to the correct level of disinfection is a high-yield topic on the CIC® exam and essential for safe infection prevention practice.
==========
Which of the following BEST describes the content of an interpretive surveillance report?
Outlines the organization’s mission for patient quality and safety
Cites the frequency of the evaluation of the monitoring program
Highlights the steps of the facility’s quality improvement activities
Provides findings in a manner designed for the intended audience
The CBIC Certified Infection Control Exam Study Guide (6th edition) explains that an interpretive surveillance report goes beyond simply presenting raw data. Its primary purpose is to translate surveillance findings into meaningful, actionable information that can be understood and used by the intended audience, such as frontline staff, clinical leaders, executive leadership, or quality committees.
Interpretive reports contextualize infection data by explaining trends, comparisons, implications, and recommended actions. This may include highlighting increases or decreases in infection rates, identifying areas of concern, interpreting statistical significance, and linking findings to prevention strategies. The format, level of detail, and language are tailored to the audience’s role and decision-making responsibilities. For example, senior leadership may need high-level summaries and risk implications, while unit-level staff benefit from detailed, practice-focused feedback.
Option A describes a mission statement, not a surveillance report. Option B refers to program evaluation logistics rather than interpretation of findings. Option C outlines quality improvement processes but does not describe how surveillance data are communicated.
For the CIC® exam, it is essential to recognize that interpretive surveillance reporting focuses on meaningful communication, not just data display. Providing findings in a manner designed for the intended audience ensures surveillance data drive prevention actions, accountability, and performance improvement—making option D the best answer.
A microbiology laboratory plays a pivotal role in both endemic and epidemic epidemiology. Which of the following should be investigated FIRST?
One blood isolate of Streptococcus agalactiae in the nursery.
Two isolates of Staphylococcus aureus in postoperative surgical sites.
Three respiratory isolates of multi-drug resistant Klebsiella pneumoniae in the medical ICU.
Two blood isolates of coagulase-negative staphylococci in the oncology unit.
Multi-drug resistant (MDR) Klebsiella pneumoniae in a high-risk area like the ICU requires urgent investigation because:
It spreads rapidly via contaminated hands or equipment.
It poses a serious risk to immunocompromised patients.
An outbreak could lead to severe hospital-acquired infections (HAIs).
Why the Other Options Are Incorrect?
A. One blood isolate of Streptococcus agalactiae in the nursery – Single cases are not indicative of an outbreak.
B. Two isolates of Staphylococcus aureus in postoperative surgical sites – Common post-surgical pathogen; requires monitoring but not immediate outbreak investigation.
D. Two blood isolates of coagulase-negative staphylococci in the oncology unit – Common contaminants in blood cultures and not immediately alarming.
CBIC Infection Control Reference
APIC guidelines prioritize investigating MDR pathogens in high-risk units, such as ICU, to prevent transmission.
A 22-year-old male has a splenectomy secondary to trauma. Which of the following vaccines is MOST important for this patient?
Haemophilus influenzae type B
Pneumococcal
Hepatitis B
Varicella
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that patients who have undergone splenectomy are at significantly increased risk for overwhelming postsplenectomy infection (OPSI), a rapidly progressive and potentially fatal condition. The spleen plays a critical role in clearing encapsulated organisms, and its absence markedly increases susceptibility to infections caused by these pathogens.
Among encapsulated bacteria, Streptococcus pneumoniae is the most common and most deadly cause of OPSI, making pneumococcal vaccination the single most important immunization for asplenic patients. Pneumococcal disease in individuals without a spleen can progress rapidly to sepsis, meningitis, or death, even in young and otherwise healthy adults. Therefore, ensuring pneumococcal vaccination—using the appropriate conjugate and polysaccharide vaccines according to age and immunization history—is a top priority.
While Haemophilus influenzae type B (Option A) and meningococcal vaccines are also recommended for asplenic patients, pneumococcal vaccination provides the greatest immediate protection against the most common cause of severe infection. Hepatitis B (Option C) and varicella (Option D) are important routine immunizations but are not specifically related to the increased infection risk associated with asplenia.
For the CIC® exam, it is critical to recognize that loss of splenic function necessitates prioritization of vaccines targeting encapsulated organisms, with pneumococcal vaccination being the most important.
A surgical team is performing a liver transplant. Which of the following represents the HIGHEST risk for transmission of a healthcare-associated infection?
Failure to change surgical gloves after contamination.
Using alcohol-based hand rub instead of surgical scrub.
Delayed administration of preoperative antibiotics.
Airflow disruption due to personnel movement.
Glove Contamination and SSI Risk:
Failure to change contaminated gloves increases the risk of surgical site infections (SSIs).
Double-gloving with an outer glove change reduces contamination.
Why Other Options Are Incorrect:
B. Alcohol-based hand rubs: Are FDA-approved alternatives to traditional scrubs and effective.
C. Delayed antibiotics: Increases infection risk, but immediate correction reduces harm.
D. Airflow disruption: Can increase SSI risk, but glove contamination poses a more direct threat.
CBIC Infection Control References:
APIC-JCR Workbook, "Surgical Infection Prevention," Chapter 6.
Which of the following microorganisms does NOT cause gastroenteritis in humans?
Norovirus
Rhinovirus
Rotavirus
Coxsackievirus
Gastroenteritis, characterized by inflammation of the stomach and intestines, typically presents with symptoms such as diarrhea, vomiting, and abdominal pain. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes the identification of infectious agents in the "Identification of Infectious Disease Processes" domain, aligning with the Centers for Disease Control and Prevention (CDC) guidelines on foodborne and enteric diseases. The question requires identifying the microorganism among the options that does not cause gastroenteritis, necessitating an evaluation of each pathogen’s clinical associations.
Option B, "Rhinovirus," is the correct answer as it does not cause gastroenteritis. Rhinoviruses are the primary cause of the common cold, affecting the upper respiratory tract and leading to symptoms like runny nose, sore throat, and cough. The CDC and WHO classify rhinoviruses as picornaviruses that replicate in the nasopharynx, with no significant evidence linking them to gastrointestinal illness in humans. Their transmission is primarily through respiratory droplets, not the fecal-oral route associated with gastroenteritis.
Option A, "Norovirus," is a well-known cause of gastroenteritis, often responsible for outbreaks of acute vomiting and diarrhea, particularly in closed settings like cruise ships or nursing homes. The CDC identifies norovirus as the leading cause of foodborne illness in the U.S., transmitted via the fecal-oral route. Option C, "Rotavirus," is a major cause of severe diarrheal disease in infants and young children worldwide, also transmitted fecal-orally, with the CDC noting its significance before widespread vaccination reduced its impact. Option D, "Coxsackievirus," a member of the enterovirus genus, can cause gastroenteritis, particularly in children, alongside other syndromes like hand-foot-mouth disease. The CDC and clinical literature (e.g., Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases) document its gastrointestinal involvement, though it is less common than norovirus or rotavirus.
The CBIC Practice Analysis (2022) and CDC guidelines on enteric pathogens underscore the importance of distinguishing between respiratory and gastrointestinal pathogens for effective infection control. Rhinovirus’s exclusive association with respiratory illness makes Option B the microorganism that does not cause gastroenteritis.
In which of the following ways is human immunodeficiency virus similar to the Hepatitis B virus?
The primary mechanism of transmission for both is maternal-fetal
Needlestick exposure leads to a high frequency of healthcare worker infection
Transmission may occur from asymptomatic carriers
The risk of infection from mucous membrane exposure is the same
The human immunodeficiency virus (HIV) and Hepatitis B virus (HBV) are both bloodborne pathogens that pose significant risks in healthcare settings, and understanding their similarities is crucial for infection prevention and control. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes the importance of recognizing transmission modes and implementing appropriate precautions in the "Prevention and Control of Infectious Diseases" domain, aligning with guidelines from the Centers for Disease Control and Prevention (CDC). Comparing these viruses involves evaluating their epidemiology, transmission routes, and occupational risks.
Option C, "Transmission may occur from asymptomatic carriers," is the correct answer. Both HIV and HBV can be transmitted by individuals who are infected but show no symptoms, making asymptomatic carriage a significant similarity. For HBV, chronic carriers (estimated at 257 million globally per WHO, 2019) can transmit the virus through blood, semen, or other bodily fluids without overt signs of disease. Similarly, HIV-infected individuals can remain asymptomatic for years during the latent phase, yet still transmit the virus through sexual contact, blood exposure, or perinatal transmission. The CDC’s "Guidelines for Prevention of Transmission of HIV and HBV to Healthcare Workers" (1987, updated 2011) and "Epidemiology and Prevention of Viral Hepatitis" (2018) highlight this shared characteristic, underscoring the need for universal precautions regardless of symptom status.
Option A, "The primary mechanism of transmission for both is maternal-fetal," is incorrect. While maternal-fetal transmission (perinatal transmission) is a significant route for both HIV and HBV—occurring in 5-10% of cases without intervention for HBV and 15-45% for HIV without antiretroviral therapy—it is not the primary mechanism. For HBV, the primary mode is horizontal transmission through unprotected sexual contact or percutaneous exposure (e.g., needlesticks), accounting for the majority of cases. For HIV, sexual transmission and intravenous drug use are the leading modes globally, with maternal-fetal transmission being a smaller proportion despite its importance. Option B, "Needlestick exposure leads to a high frequency of healthcare worker infection," is partially true but not a precise similarity. Needlestick exposures carry a high risk for HBV (transmission risk ~30% if the source is HBeAg-positive) and a lower risk for HIV (~0.3%), but the frequency of infection among healthcare workers is significantly higher for HBV due to its greater infectivity and stability outside the host. This makes the statement more characteristic of HBV than a shared trait. Option D, "The risk of infection from mucous membrane exposure is the same," is false. The risk of HIV transmission via mucous membrane exposure (e.g., splash to eyes or mouth) is approximately 0.09%, while for HBV it is higher (up to 1-2% depending on viral load and exposure type), reflecting HBV’s greater infectivity.
The CBIC Practice Analysis (2022) and CDC guidelines emphasize the role of asymptomatic transmission in shaping infection control strategies, such as routine testing and post-exposure prophylaxis. This shared feature of HIV and HBV justifies Option C as the most accurate similarity.
Which of the following processes is essential for endoscope reprocessing?
Intermediate level disinfection and contact time
Pre-cleaning, leak testing, and manual cleaning
Inspection using a borescope and horizontal storage
Leak testing, manual cleaning, and low level disinfection
The correct answer is B, "Pre-cleaning, leak testing, and manual cleaning," as these processes are essential for endoscope reprocessing. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, proper reprocessing of endoscopes is critical to prevent healthcare-associated infections (HAIs), given their complex design and susceptibility to microbial contamination. The initial steps of pre-cleaning (removing gross debris at the point of use), leak testing (ensuring the endoscope’s integrity to prevent fluid ingress), and manual cleaning (using enzymatic detergents to remove organic material) are foundational to the reprocessing cycle. These steps prepare the endoscope for high-level disinfection or sterilization by reducing bioburden and preventing damage, as outlined in standards such as AAMI ST91 (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). Failure at this stage can compromise subsequent disinfection, making it a non-negotiable component of the process.
Option A (intermediate level disinfection and contact time) is an important step but insufficient alone, as intermediate-level disinfection does not achieve the high-level disinfection required for semi-critical devices like endoscopes, which must eliminate all microorganisms except high levels of bacterial spores. Option C (inspection using a borescope and horizontal storage) includes valuable quality control (inspection) and storage practices, but these occur later in the process and are not essential initial steps; vertical storage is often preferred to prevent damage. Option D (leak testing, manual cleaning, and low level disinfection) includes two essential steps (leak testing and manual cleaning) but is inadequate because low-level disinfection does not meet the standard for endoscopes, which require high-level disinfection or sterilization.
The emphasis on pre-cleaning, leak testing, and manual cleaning aligns with CBIC’s focus on adhering to evidence-based reprocessing protocols to ensure patient safety and prevent HAIs (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.4 - Implement environmental cleaning and disinfection protocols). These steps are mandated by guidelines to mitigate risks associated with endoscope use in healthcare settings.
An infection preventionist is providing education to a group of medical device reprocessing staff on critical steps in cleaning instruments. Which of the following actions is recommended while using washer-disinfector?
Stack instruments inside the machine
Use circulating water with a pH of 3
Disassemble instruments as much as possible
Close hinged instruments prior to placing in the machine
Best practices for using a washer-disinfector include disassembling instruments and opening hinged instruments to ensure proper cleaning and decontamination.
The APIC Text explains:
“Open hinged instruments and disassemble all instruments… Confirm that spray will be able to reach all loaded items without impedance.”
This ensures water and detergents reach all surfaces. Avoid stacking instruments and ensure proper placement to allow full cleaning.
An infection preventionist (IP) is asked to assist in rewriting policies for insertion and maintenance of IV catheters. Which of the following are acceptable for use in site preparation?
Chloroxylenol or acetone
Povidone-iodine or para-chloro-meta-xylenol (PCMX)
Alcohol or chlorhexidine
Benzalkonium chloride or chlorhexidine
For IV catheter insertion, evidence-based guidance recommends preparing skin with an effective antiseptic agent to reduce skin flora at the insertion site and lower catheter-related infection risk. CDC guidance for prevention of intravascular catheter-related infections specifies that clean skin should be prepared with >0.5% chlorhexidine (CHG) in alcohol for central venous catheter and peripheral arterial catheter insertion and during dressing changes. If CHG is contraindicated, CDC lists tincture of iodine, an iodophor, or 70% alcohol as acceptable alternatives.
Option C (Alcohol or chlorhexidine) is the only answer in which both agents are recognized as appropriate antiseptics for site preparation in intravascular catheter guidance (alcohol as an acceptable antiseptic option; CHG as preferred, typically in alcohol).
The other choices include agents that are not recommended as standard site-prep antiseptics for catheter insertion in major guidelines: acetone is not an antiseptic for vascular access site prep; benzalkonium chloride is generally considered less effective for this purpose compared with CHG/alcohol/iodophors; and PCMX/chloroxylenol is not the typical recommended agent for catheter insertion site antisepsis in these guidelines.
When assessing a patient’s infection prevention and control educational needs, it is necessary to FIRST determine the patient’s
severity of illness.
educational background.
duration of hospitalization.
baseline knowledge of the subject.
The correct answer is D, "baseline knowledge of the subject," as this is the necessary first step when assessing a patient’s infection prevention and control educational needs. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, effective patient education in infection prevention and control requires a tailored approach that begins with understanding the patient’s existing knowledge and comprehension of the topic. Determining baseline knowledge allows the infection preventionist (IP) to identify gaps, customize educational content to the patient’s level of understanding, and ensure the information is relevant and actionable (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.1 - Develop and implement educational programs). This step ensures that education is neither too basic nor overly complex, maximizing its effectiveness in promoting behaviors such as hand hygiene, wound care, or adherence to isolation protocols.
Option A (severity of illness) is an important clinical consideration that may influence the timing or method of education delivery, but it is not the first step in assessing educational needs. The severity might affect the patient’s ability to learn, but it does not directly inform the content or starting point of the education. Option B (educational background) provides context about the patient’s general learning capacity (e.g., literacy level or language preference), but it is secondary to assessing specific knowledge about infection prevention, as background alone does not reveal current understanding. Option C (duration of hospitalization) may impact the opportunity for education but is not a primary factor in determining what the patient needs to learn; it is more relevant to scheduling or prioritizing educational interventions.
The focus on baseline knowledge aligns with adult learning principles endorsed by CBIC, which emphasize assessing learners’ prior knowledge to build effective educational strategies (CBIC Practice Analysis, 2022, Domain IV: Education and Research, Competency 4.2 - Evaluate the effectiveness of educational programs). This approach ensures patient-centered care and supports infection control by empowering patients with the knowledge to participate in their own prevention efforts.
One of the elements of antibiotic stewardship is controlling antibiotic use. Which of the following BEST describes a closed formulary?
Requires the prescribing physician to obtain some form of approval before the antibiotic will be dispensed.
Automatic switching from broad-spectrum empiric therapy to narrower-spectrum agents.
Practice of rotating two or more classes of formulary drugs on a regular basis.
Limits the number of antibiotics available for physicians to prescribe.
Antibiotic stewardship programs are designed to optimize antimicrobial use, improve patient outcomes, reduce antimicrobial resistance, and decrease unnecessary costs. The CBIC Certified Infection Control Exam Study Guide (6th edition) identifies formulary restriction and preauthorization as key core strategies within effective antimicrobial stewardship programs. A closed formulary specifically refers to a system in which access to certain antibiotics is restricted and requires prior approval before dispensing.
In a closed formulary model, prescribers must obtain authorization—often from infectious diseases specialists, pharmacy, or an antimicrobial stewardship team—before selected antimicrobial agents can be used. This approach ensures that high-risk, broad-spectrum, or high-cost antibiotics are used only when clinically appropriate. By requiring approval, the organization promotes judicious antibiotic selection, prevents unnecessary exposure, and supports resistance prevention efforts.
Option B describes de-escalation, which is another stewardship strategy but does not define a closed formulary. Option C refers to antibiotic cycling, a controversial and less-supported strategy. Option D is incorrect because a closed formulary does not merely limit availability; rather, it controls access through approval mechanisms.
For the CIC® exam, it is critical to distinguish between stewardship strategies. A closed formulary is best characterized by mandatory approval prior to dispensing, making option A the most accurate answer according to the Study Guide’s antimicrobial stewardship framework.
==========
The degree of infectiousness of a patient with tuberculosis correlates with
the hand-hygiene habits of the patient.
a presence of acid-fast bacilli in the blood.
a tuberculin skin test result that is greater than 20 mm
the number of organisms expelled into the air
The infectiousness of tuberculosis (TB) is directly related to the number of Mycobacterium tuberculosis organisms expelled into the air by an infected patient.
Step-by-Step Justification:
TB Transmission Mechanism:
TB spreads through airborne droplet nuclei, which remain suspended for long periods.
Factors Affecting Infectiousness:
High bacterial load in sputum: Smear-positive patients are much more infectious.
Coughing and sneezing frequency: More expelled droplets increase exposure risk.
Environmental factors: Poor ventilation increases transmission.
Why Other Options Are Incorrect:
A. Hand hygiene habits: TB is airborne, not transmitted via hands.
B. Presence of acid-fast bacilli (AFB) in blood: TB is not typically hematogenous, and blood AFB does not correlate with infectiousness.
C. Tuberculin skin test (TST) >20 mm: TST indicates prior exposure, not infectiousness.
CBIC Infection Control References:
APIC Text, "Tuberculosis Transmission and Control Measures".
Which of the following represents the most effective strategy for preventing Clostridioides difficile transmission in a healthcare facility?
Daily environmental cleaning with quaternary ammonium compounds.
Strict antimicrobial stewardship to limit unnecessary antibiotic use.
Universal C. difficile screening on admission for high-risk patients.
Routine use of alcohol-based hand rub for hand hygiene after patient contact.
Antimicrobial stewardship is the most effective strategy to reduce C. difficile infections (CDI) by limiting the use of broad-spectrum antibiotics.
Quaternary ammonium disinfectants (A) are ineffective against C. difficile spores; bleach-based disinfectants are preferred.
Routine screening (C) is not cost-effective for prevention.
Alcohol-based hand rubs (D) do not kill C. difficile spores; soap and water should be used.
CBIC Infection Control References:
APIC Text, "C. difficile Prevention Strategies," Chapter 9.
What inflammatory reaction may occur in the eye after cataract surgery due to a breach in disinfection and sterilization of intraocular surgical instruments?
Endophthalmitis
Bacterial conjunctivitis
Toxic Anterior Segment Syndrome
Toxic Posterior Segment Syndrome
The correct answer is C, "Toxic Anterior Segment Syndrome," as this is the inflammatory reaction that may occur in the eye after cataract surgery due to a breach in disinfection and sterilization of intraocular surgical instruments. According to the Certification Board of Infection Control and Epidemiology (CBIC) guidelines, Toxic Anterior Segment Syndrome (TASS) is a sterile, acute inflammatory reaction that can result from contaminants introduced during intraocular surgery, such as endotoxins, residues from improper cleaning, or chemical agents left on surgical instruments due to inadequate disinfection or sterilization processes (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.3 - Ensure safe reprocessing of medical equipment). TASS typically presents within 12-48 hours post-surgery with symptoms like pain, redness, and anterior chamber inflammation, and it is distinct from infectious causes because it is not microbial in origin. A breach in reprocessing protocols, such as failure to remove detergents or improper sterilization, is a known risk factor, making it highly relevant to infection prevention efforts in surgical settings.
Option A (endophthalmitis) is an infectious inflammation of the internal eye structures, often caused by bacterial or fungal contamination, which can also result from poor sterilization but is distinguished from TASS by its infectious nature and longer onset (days to weeks). Option B (bacterial conjunctivitis) affects the conjunctiva and is typically a surface infection unrelated to intraocular surgery or sterilization breaches of surgical instruments. Option D (toxic posterior segment syndrome) is not a recognized clinical entity in the context of cataract surgery; inflammation in the posterior segment is more commonly associated with infectious endophthalmitis or other conditions, not specifically linked to reprocessing failures.
The focus on TASS aligns with CBIC’s emphasis on ensuring safe reprocessing to prevent adverse outcomes in surgical patients, highlighting the need for rigorous infection control measures (CBIC Practice Analysis, 2022, Domain III: Infection Prevention and Control, Competency 3.5 - Evaluate the environment for infection risks). This is supported by CDC and American Academy of Ophthalmology guidelines, which identify TASS as a preventable complication linked to reprocessing errors (CDC Guidelines for Disinfection and Sterilization, 2019; AAO TASS Task Force Report, 2017).
The Sterile Processing Deportment alerts an infection preventionist that a load of surgical Instruments sterilized with high temperature steam:moist heat needs to be recalled. Which of the following Is the MOST likely reason for the recall?
Failure of the biological Indicator Bacillus subtilts
Failure of the biological Indicator Geobaciltus stearothermophilus
Placement of the biological Indicator on the bottom shelf over the d*an
Incorrect placement of the instruments In the tray
The most likely reason for the recall of a steam-sterilized load is the failure of the biological indicator (BI), specifically Geobacillus stearothermophilus, which is used to monitor high-temperature steam (moist heat) sterilization processes. This organism is the biological indicator of choice because it has high resistance to moist heat and thus serves as a reliable marker for sterilization efficacy.
The APIC Text and AAMI ST79 guidelines confirm that Geobacillus stearothermophilus is used for steam sterilization and that a failed BI indicates a failure in the sterilization process, which requires immediate action, including recalling all items sterilized since the last negative BI and reprocessing them. This is a crucial aspect of ensuring patient safety and preventing the use of potentially non-sterile surgical instruments.
According to the APIC Text:
"BIs are the only process indicators that directly monitor the lethality of a given sterilization process. [...] Geobacillus stearothermophilus spores are used to monitor steam sterilization..."
The CIC Study Guide (6th ed.) also specifies that:
"Evidence of sterilization failures (e.g., positive biological indicators) is the most common reason for a recall."
Additionally, it is noted:
“With steam sterilization, the instrument load does not need to be recalled for a single positive biological indicator test, with the exception of implantable objects.”
However, multiple positive BIs or BI failure confirmation does require a recall.
The incorrect options explained:
A. Bacillus subtilis – This is not used in steam sterilization but rather in dry heat or EO processes.
C. Placement of the biological indicator on the bottom shelf over the drain – While incorrect placement can lead to test failure, the recall is prompted by BI failure, not just placement.
D. Incorrect placement of instruments – This can cause sterilization failure but is not the direct trigger for a recall unless it leads to a failed BI.
Which of the following is the correct collection technique to obtain a laboratory specimen for suspected pertussis?
Cough plate
Nares culture
Sputum culture
Nasopharyngeal culture
The gold standard specimen for diagnosing pertussis (Bordetella pertussis infection) is a nasopharyngeal culture because:
B. pertussis colonizes the nasopharynx, making it the best site for detection.
A properly collected nasopharyngeal swab or aspirate increases diagnostic sensitivity.
This method is recommended for culture, PCR, or direct fluorescent antibody testing.
Why the Other Options Are Incorrect?
A. Cough plate – Not commonly used due to low sensitivity.
B. Nares culture – The nares are not a primary site for pertussis colonization.
C. Sputum culture – B. pertussis does not commonly infect the lower respiratory tract.
CBIC Infection Control Reference
APIC confirms that nasopharyngeal culture is the preferred method for diagnosing pertussis.
An infection preventionist (IP) observes an increase in primary bloodstream infections in patients admitted through the Emergency Department. Poor technique is suspected when peripheral intravenous (IV) catheters are inserted. The IP should FIRST stratify infections by:
Location of IV insertion: pre-hospital, Emergency Department, or in-patient unit.
Type of dressing used: gauze, CHG impregnated sponge, or transparent.
Site of insertion: hand, forearm, or antecubital fossa.
Type of skin preparation used for the IV site: alcohol, CHG/alcohol, or iodophor.
When an infection preventionist (IP) identifies an increase in primary bloodstream infections (BSIs) associated with peripheral intravenous (IV) catheter insertion, the initial step in outbreak investigation and process improvement is to stratify the data to identify potential sources or patterns of infection. According to the Certification Board of Infection Control and Epidemiology (CBIC), the "Surveillance and Epidemiologic Investigation" domain emphasizes the importance of systematically analyzing data to pinpoint contributing factors, such as location, technique, or equipment use, in healthcare-associated infections (HAIs). The question specifies poor technique as a suspected cause, and the first step should focus on contextual factors that could influence technique variability.
Option A, stratifying infections by the location of IV insertion (pre-hospital, Emergency Department, or in-patient unit), is the most logical first step. Different settings may involve varying levels of training, staffing, time pressure, or adherence to aseptic technique, all of which can impact infection rates. For example, pre-hospital settings (e.g., ambulance services) may have less controlled environments or less experienced personnel compared to in-patient units, potentially leading to technique inconsistencies. The CDC’s Guidelines for the Prevention of Intravascular Catheter-Related Infections (2017) recommend evaluating the context of catheter insertion as a critical initial step in investigating BSIs, making this a priority for the IP to identify where the issue is most prevalent.
Option B, stratifying by the type of dressing used (gauze, CHG impregnated sponge, or transparent), is important but should follow initial location-based analysis. Dressings play a role in maintaining catheter site integrity and preventing infection, but their impact is secondary to the insertion technique itself. Option C, stratifying by the site of insertion (hand, forearm, or antecubital fossa), is also relevant, as anatomical sites differ in infection risk (e.g., the hand may be more prone to contamination), but this is a more specific factor to explore after broader contextual data is assessed. Option D, stratifying by the type of skin preparation used (alcohol, CHG/alcohol, or iodophor), addresses antiseptic efficacy, which is a key component of technique. However, without first understanding where the insertions occur, it’s premature to focus on skin preparation alone, as technique issues may stem from systemic factors across locations.
The CBIC Practice Analysis (2022) supports a stepwise approach to HAI investigation, starting with broad stratification (e.g., by location) to guide subsequent detailed analysis (e.g., technique-specific factors). This aligns with the CDC’s hierarchical approach to infection prevention, where contextual data collection precedes granular process evaluation. Therefore, the IP should first stratify by location to establish a baseline for further investigation.
After reviewing quarterly surveillance data, the infection preventionist (IP) identifies an upward trend in needlestick injuries occurring during bedside blood draws. What is the MOST appropriate collaborative action for the IP to take with the Occupational Health Department?
Wait for the next regulatory survey to address the issue more formally with hospital leadership.
Complete a device review and determine if faulty equipment has been the cause or if the manufacturer has issued a recall.
Convene a multidisciplinary safety team to evaluate workflows, review device usage, and recommend practice changes.
Discuss trends at the quarterly quality assurance and performance improvement meeting with the multidisciplinary safety team.
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that when surveillance identifies an increasing trend in occupational exposures, such as needlestick injuries, the infection preventionist must take prompt, proactive, and collaborative action. The most appropriate response is to convene a multidisciplinary safety team in partnership with Occupational Health to perform a focused evaluation of the problem.
A multidisciplinary approach allows for comprehensive assessment of workflows, staffing practices, device selection, training, and compliance with standard precautions. Team members may include nursing leadership, frontline staff, occupational health, infection prevention, materials management, and safety officers. This collaboration supports root cause analysis to identify contributing factors—such as improper technique, workflow inefficiencies, inadequate training, or suboptimal safety-engineered devices—and to implement targeted interventions.
Option A is inappropriate because delaying action increases risk to healthcare personnel. Option B may be part of the evaluation but is too narrow and should not occur in isolation. Option D is insufficient because discussing trends alone does not result in immediate corrective action.
The Study Guide highlights that timely, interdisciplinary performance improvement efforts are essential to reduce occupational exposures and comply with regulatory and safety standards. Convening a multidisciplinary safety team enables rapid intervention, staff engagement, and sustainable injury reduction—making option C the best answer and a high-yield CIC® exam concept.
==========
A suspected measles case has been identified in an outpatient clinic without an airborne infection isolation room (AIIR). Which of the following is the BEST course of action?
Patient should be sent home
Staff should don a respirator, gown, and face shield.
Patient should be offered the Measles. Mumps, Rubella (MMR) vaccine
Patient should be masked and placed in a private room with door closed.
Measles is a highly contagious airborne disease, and the best immediate action in an outpatient clinic without an Airborne Infection Isolation Room (AIIR) is to mask the patient and isolate them in a private room with the door closed.
Why the Other Options Are Incorrect?
A. Patient should be sent home – While home isolation may be necessary, sending the patient home without proper precautions increases exposure risk.
B. Staff should don a respirator, gown, and face shield – While N95 respirators are necessary for staff, this does not address patient containment.
C. Patient should be offered the MMR vaccine – The vaccine does not treat active measles infection and should be given only as post-exposure prophylaxis to susceptible contacts.
CBIC Infection Control Reference
Measles cases in outpatient settings require immediate airborne precautions to prevent transmission.
A patient with a non-crusted rash has boon diagnosed with Sarcoptes scabiei. The patient is treated with 5% permethrin and precautions are started. The precautions can be stopped
when the treatment cream is applied
when the bed linen is changed
24 hours after effective treatment
24 hours after the second treatment
For Sarcoptes scabiei (scabies), Contact Precautions should remain in place until 24 hours after effective treatment has been completed. The first-line treatment is 5% permethrin cream, which is applied to the entire body and left on for 8–14 hours before being washed off.
Why the Other Options Are Incorrect?
A. When the treatment cream is applied – The mite is still present and infectious until treatment has fully taken effect.
B. When the bed linen is changed – While changing linens is necessary, it does not indicate that the infestation has cleared.
D. 24 hours after the second treatment – Most cases require only one treatment with permethrin, though severe cases may need a second dose after a week.
CBIC Infection Control Reference
According to APIC guidelines, Contact Precautions can be discontinued 24 hours after effective treatment has been administered.
Which of the following BEST reduces the risk of infection in patients undergoing peritoneal dialysis?
Daily dressing change of the catheter exit site
Weekly surveillance cultures of catheter exit sites
Catheter insertion in the operating room
Irrigation of catheter with antimicrobials between dialysis treatments
The CBIC Certified Infection Control Exam Study Guide (6th edition) emphasizes that the most effective strategy for reducing infection risk in peritoneal dialysis (PD) patients is ensuring optimal conditions at the time of catheter insertion. Placement of the peritoneal dialysis catheter in the operating room provides a controlled, sterile environment that minimizes microbial contamination and significantly reduces the risk of early peritonitis and exit-site infections.
Peritoneal dialysis–associated infections are most often linked to contamination during catheter insertion or manipulation. Performing catheter insertion in the operating room allows for strict adherence to aseptic technique, appropriate airflow controls, surgical hand antisepsis, and use of sterile instruments—all of which are essential infection prevention measures highlighted in the Study Guide.
The other options are less effective or not recommended. Daily dressing changes (Option A) may actually increase manipulation of the exit site and raise infection risk if not clinically indicated. Weekly surveillance cultures (Option B) are not recommended, as they do not prevent infection and may lead to unnecessary antimicrobial use. Irrigating catheters with antimicrobials (Option D) is discouraged because it has not been shown to reduce infection rates and may contribute to antimicrobial resistance.
For the CIC® exam, it is important to recognize that prevention of peritoneal dialysis–associated infection begins with proper catheter placement under optimal sterile conditions, making operating room insertion the best answer.
==========
An infection control manager is training a new infection preventionist. In discussing surveillance strategies, which of the following types of hospital infection surveillance usually provides maximum benefit with minimum resources?
High-risk patient focus
Antibiotic monitoring
Prevalence surveys
Nursing care plan review
A high-risk patient focus maximizes benefits while minimizing resource use in infection surveillance.
Step-by-Step Justification:
Efficiency of High-Risk Surveillance:
Targeting ICU, immunocompromised patients, or surgical units helps detect infections where the risk is highest, leading to earlier interventions.
Resource Allocation:
Full hospital-wide surveillance is resource-intensive; focusing on high-risk groups is more efficient.
Why Other Options Are Incorrect:
B. Antibiotic monitoring:
Important for stewardship, but not the primary focus of infection surveillance.
C. Prevalence surveys:
Snapshot data only; does not provide ongoing monitoring.
D. Nursing care plan review:
Less direct in identifying infection trends.
CBIC Infection Control References:
APIC Text, "Surveillance Strategies for Infection Prevention".
Some pathogens live in the body and can be cultured, but do NOT elicit any response from the body’s defense mechanisms. This state is called:
Colonization
Infection
Latency
Contamination
The interaction between pathogens and the human body can take various forms, each with distinct immunological and clinical implications. The Certification Board of Infection Control and Epidemiology (CBIC) emphasizes understanding these states within the "Identification of Infectious Disease Processes" domain to guide infection prevention strategies. The question describes a scenario where pathogens are present, can be cultured (indicating viable organisms), but do not trigger a response from the body’s defense mechanisms, such as inflammation or immune activation. This requires identifying the appropriate microbiological state.
Option A, "Colonization," is the correct answer. Colonization occurs when microorganisms are present on or in the body (e.g., skin, mucous membranes, or gut) without causing harm or eliciting an immune response. These pathogens can be cultured, as they are alive and replicating, but they exist in a commensal or symbiotic relationship with the host, not provoking symptoms or defense mechanisms. Examples include normal flora like Staphylococcus epidermidis on the skin or Streptococcus salivarius in the oral cavity. The Centers for Disease Control and Prevention (CDC) defines colonization as the presence of microbes without tissue invasion or damage, distinguishing it from infection (CDC, "Principles of Epidemiology in Public Health Practice," 3rd Edition, 2012).
Option B, "Infection," is incorrect because it involves the invasion and multiplication of pathogens in body tissues, leading to an immune response, such as inflammation, fever, or antibody production. This contrasts with the question’s description of no defense mechanism response. Option C, "Latency," refers to a state where a pathogen (e.g., herpes simplex virus or Mycobacterium tuberculosis) remains dormant in the body after initial infection, capable of reactivation but not eliciting an active immune response during dormancy. However, latency implies a prior infection with a latent phase, whereas the question suggests a current, non-responsive state without prior infection context. Option D, "Contamination," describes the unintended presence of pathogens on inanimate objects or surfaces (e.g., medical equipment), not within the body, and does not align with the scenario of living, culturable pathogens in a host.
The CBIC Practice Analysis (2022) and CDC guidelines highlight colonization as a key concept in infection control, particularly in settings like hospitals where colonized patients can serve as reservoirs for potential infections. The absence of an immune response, as specified, aligns with the definition of colonization, making Option A the most accurate answer.


TESTED 30 May 2026

